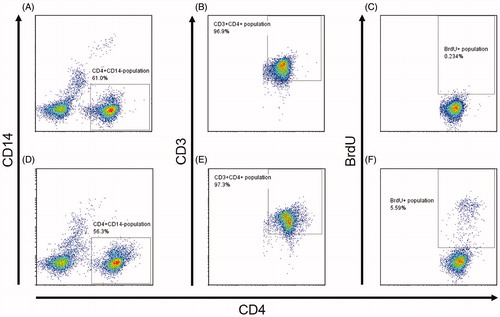
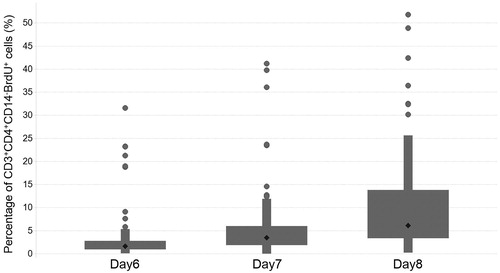
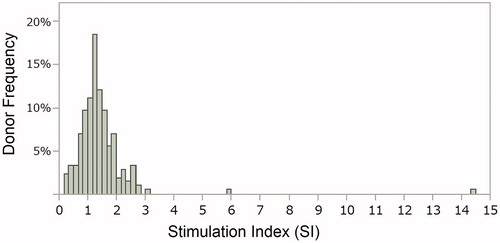
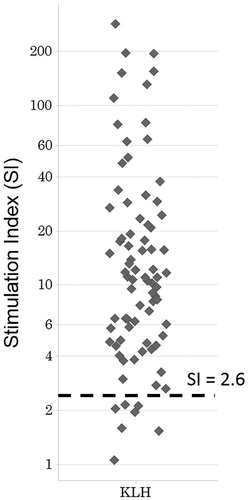
Figures & data
Table 1. Estimated sample size for statistical significance of positive response.
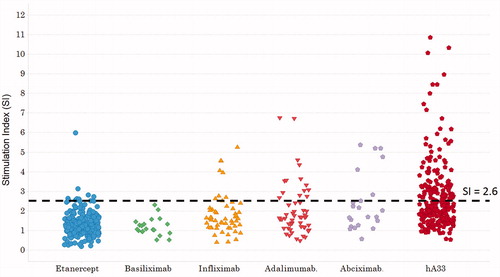
Table 2. Frequency of positive samples for each mAb in the TH cell assay.
Table 3. Clinical ADA incidence of mAb drugs.