Figures & data
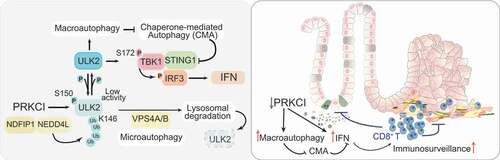
Figure 1. PRKCI controls the autophagy-interferon crosstalk to regulate cancer immunosurveillance. (Left panel) PRKCI phosphorylates and inactivates ULK2; ULK2 activates macroautophagy that represses the degradation of STING1 by chaperone-mediated autophagy; ULK2 also phosphorylates TBK1 at its S172 which serves to activate the STING1-mediated activation of the IFN pathway through phosphorylation of IRF3; PRKCI represses ULK2 activity by phosphorylating its S150 that also triggers the K63 ubiquitination of ULK2’s K146 through the NDFIP1-NEDD4L complex, targeting ULK2 for degradation through microautophagy; activation of macroautophagy promotes the IFN cascade through inhibition of CMA. (Right panel) The inactivation of PRKCI unleashes autophagy and the IFN pathway that serves to counteract the pro-tumorigenic effect of PRKCI deficiency, restraining cancer initiation and progression.