Figures & data
Figure 1. Chemical structures of the 11 studied pyrovalerone derivatives together with the parent compounds α-PVP and MDPV.
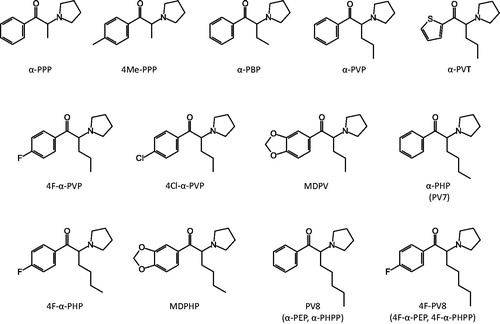
Table 1. Monoisotopic and exact masses of monitored protonated molecular ions of the 11 pyrovalerones, together with information on their appearances as recreational drugs and classification in Sweden. Information on MDPV and α-PVP is also included for comparison.
Figure 2. Chromatograms from the analysis of α-PPP in a urine sample from one selected case of intoxication. The analysis was performed with LC–HRMS in scan mode and in MS/MS mode for identification. Comparison was made with certified reference substance.
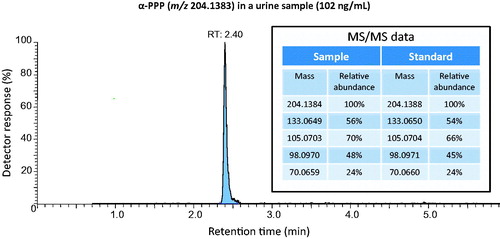
Table 2. Urine and serum concentrations of pyrovalerone NPS in intoxication cases.
Table 3. Details of the 8 single drug intoxication cases.
Figure 3. Quarterly statistics during 2010–2016 from the STRIDA project of analytically confirmed (in urine or serum) cases of intoxication involving a pyrovalerone derivative. The time for substance regulation in Sweden is indicated by a bold vertical line.
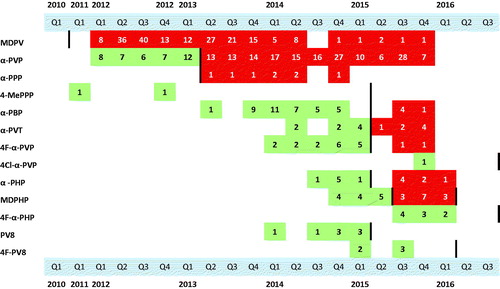
Table 4. Pyrovalerone substances mentioned in telephone inquiries to the Swedish Poisons Information Centre.
