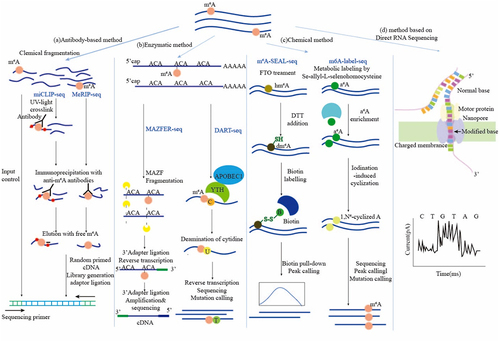
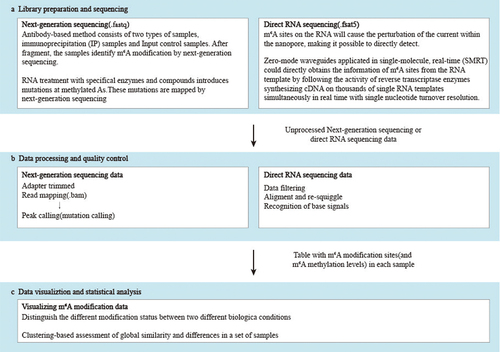
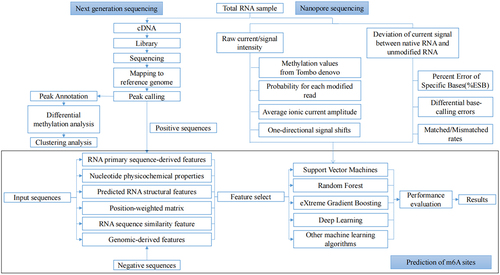
Figures & data
Table 1. Evaluation of experimental methods for m6A sites identification and quantification.
Table 2. Bioinformatics analysis tools and software based on MeRIP-seq(m6A-seq) data.
Table 3. Bioinformatics tool for nanopore sequencing.
Table 4. Summary of the reviewed predictors for m6A sites.
Table 5. m6A methylation databases.
Table 6. Validation Method for N6-Methyladenosine Sites.