Figures & data
Figure 1. Maximum projections of consecutive CLSM sections after α-tubulin immunostaining, as described in ref. 10, through the transition and elongation zones of wild-type (A) and rsw1 roots kept for 24 h at 31°C (B), or prc1 (C) roots at 22°C. Cortical microtubules are transversely oriented in transition zone cells (arrowheads) in all roots. In cells of the elongation zone (arrows), microtubule orientation is not transverse in the mutants, compared to the wild-type (B, C; cf. A). All plant materials were purchased from NASC (http://arabidopsis.info/). Scale bar: 20μm.
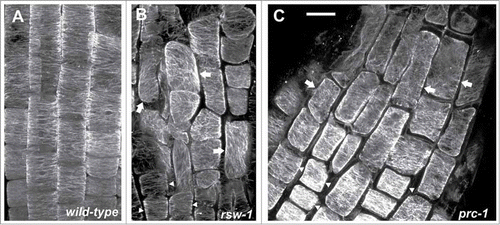
Figure 2. CLSM sections after α-tubulin immunostaining, as described in ref. 10, through the meristematic zone of wild-type (A) and rsw1 roots kept for 24 h at 31°C (B), or prc1 roots at 22°C (C). Cortical microtubules are transverse (arrows) in interphase meristematic cells. Scale bar: 20 μm.
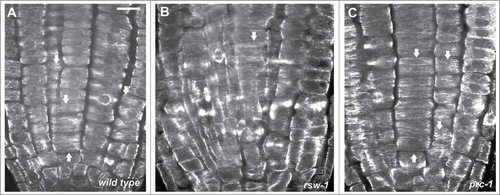
Figure 3. A graphical model illustrating the effect of cellulose-deficient primary cell wall mutants on cortical microtubule orientation in Arabidopsis thaliana root. The short primary root phenotype of pom2–4, prc1, rsw1 and than mutants is caused by reduction of anisotropic cell expansion, originating from decreased cellulose microfibril deposition. Suppression of cell expansion results in alteration of cortical microtubule orientation from a transverse (black lines) to a random (red lines) pattern in the fast elongation zone. On the contrary, in the meristematic and transition zones cortical microtubules exhibit transverse orientation (black lines) reminiscent of the pattern in wild-type roots. The mutants are: pom2–4 of Cellulose Synthase Interacting1 protein (CSI1),Citation3 prc1 (procuste1 of CesA6),Citation13 rsw1 (radially swollen1 of CesA1),Citation11,12 and than (thanatos of CesA3).Citation19