Figures & data
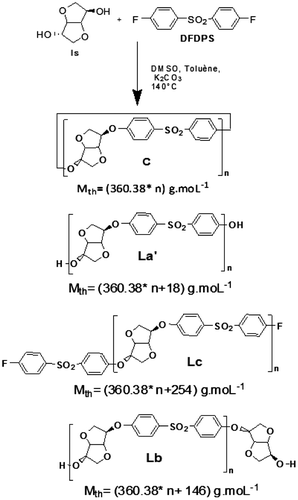
Table 1. Calculated masses (including Na+ doping) of potential structures (cyclic C; linear La’, Lb, Lc).
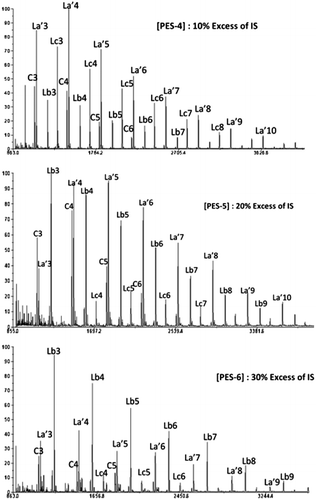
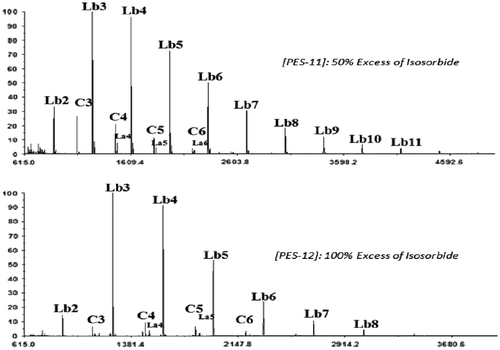
Table 2. Synthesis of PES with variation of IS excess at a concentration of 0.3 mol L−1.
Figure 4. Effect of the variation of IS excess in the end group structure of produced oligomers at a concentration of 0. 3 mol L−1.
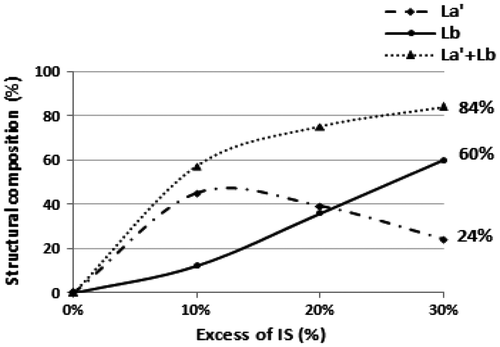
Figure 5. 1H NMR spectra [3.5–8.1 ppm] of oligoethersulfones at 0.15 mol L−1 [DMSO-D6, 500 MHz, 20 °C].
![Figure 5. 1H NMR spectra [3.5–8.1 ppm] of oligoethersulfones at 0.15 mol L−1 [DMSO-D6, 500 MHz, 20 °C].](/cms/asset/9b205f95-3c0f-4714-ae1a-475c60f8c2ee/tdmp_a_947554_f0005_b.gif)
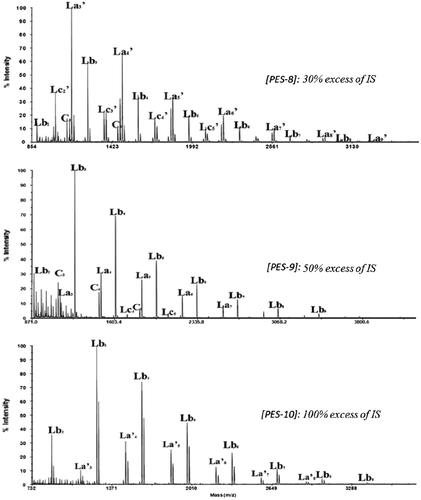
Table 3. Synthesis of PES with variation of IS excess and monomer concentration.
Figure 7. 13C-NMR spectra [68–180 ppm] of oligo-ethersulfones at 0.15 mol L−1 [DMSO-D6, 500 MHz, 20 °C].
![Figure 7. 13C-NMR spectra [68–180 ppm] of oligo-ethersulfones at 0.15 mol L−1 [DMSO-D6, 500 MHz, 20 °C].](/cms/asset/59b48d63-2720-43bf-8242-e01905c4ef92/tdmp_a_947554_f0007_b.gif)