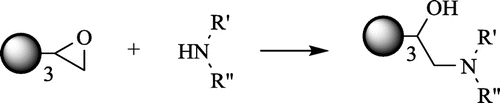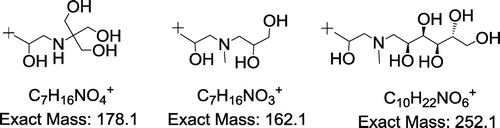Figures & data
Table 1. The amounts of reactants and solvents used in the synthesis of dendrimers 1–6.
Figure 2. The structure of the substrates I–V, used for the synthesis of dendrimers 1–6 G0 (I and II – triepoxides, III–V – aminopolyols).
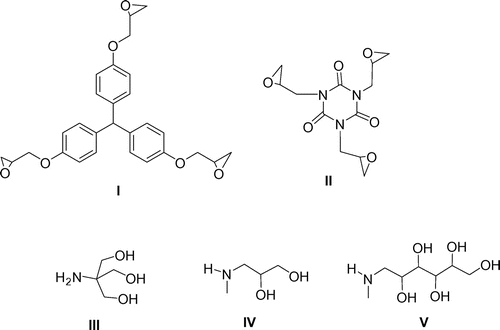
Figure 4. MALDI TOF mass spectrum of: a) a crude compound 1, matrix DHB. Ions: A = [1+H]+ i.e. [I+3Tris+H]+, m/z 824, B = [I+2Tris+H]+, m/z 703; D = [I+DHB+CH3OH+H]+, m/z 647, E =[I+Tris+H2O+H]+., m/z 599; b) purified compound 1, c) crude compound 4.
![Figure 4. MALDI TOF mass spectrum of: a) a crude compound 1, matrix DHB. Ions: A = [1+H]+ i.e. [I+3Tris+H]+, m/z 824, B = [I+2Tris+H]+, m/z 703; D = [I+DHB+CH3OH+H]+, m/z 647, E =[I+Tris+H2O+H]+., m/z 599; b) purified compound 1, c) crude compound 4.](/cms/asset/89e5fc96-d1cc-4af5-85e9-3fe31cc7017e/tdmp_a_1231048_f0004_b.gif)
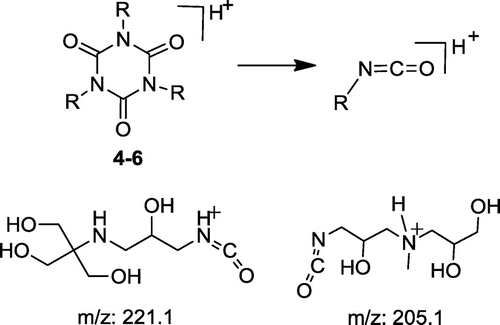
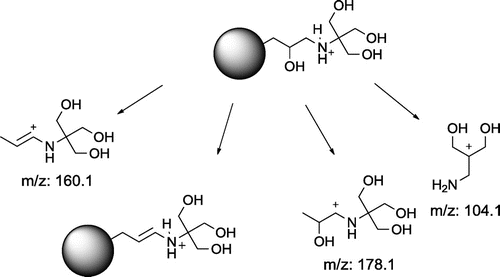
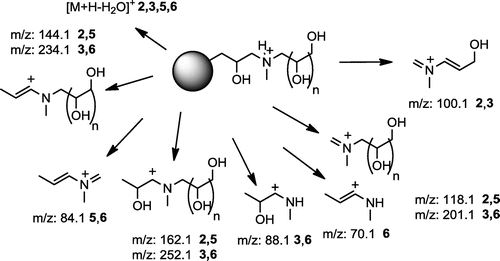
Table 2. Main ions (m/z, % r.a.) in MALDI TOF positive mass spectra of crude products 1–6 and proposed ion structures; APO = aminopolyol.
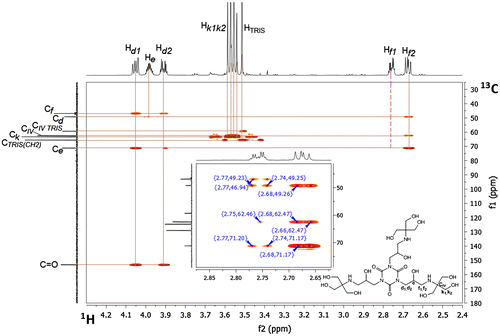
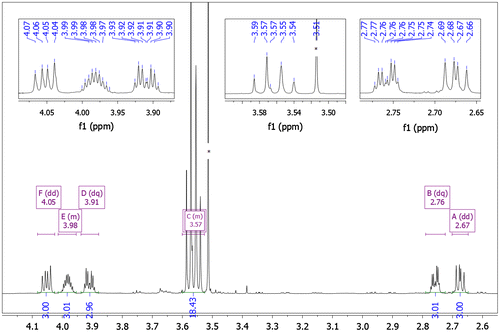
Table 3. Chemical shifts of proton (1–6) and carbon (2, 4–6) atoms δ (ppm), obtained from NMR spectra, measured in D2O; atom labeling given above the table.
Figure 6. 1H-13C HSQC NMR spectrum of compound 4 (D2O, 298 K) in the range 2.4–4.3 ppm (1H) and 30–180 ppm (13C).