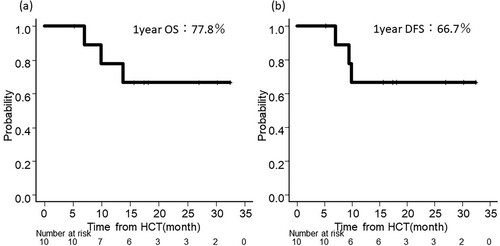
Figures & data
Figure 1. Clinical course of the 10 study patients before and after hematopoietic stem cell transplantation. Gilteritinib and quizartinib doses (mg/body) are indicated numerically. AEs were grade 3 or higher and were indicated by letters such as ‘SOS/VOD’. 
 (red): diagnosis;
(red): diagnosis;  : CR indicates complete remission;
: CR indicates complete remission;  : NR, non-remission;
: NR, non-remission;  (yellow): HCT, allogeneic hematopoietic cell transplantation;
(yellow): HCT, allogeneic hematopoietic cell transplantation;  (blue): gilteritinib; fx8(green): quizartinib;
(blue): gilteritinib; fx8(green): quizartinib;  (orange): AE, adverse event;
(orange): AE, adverse event; 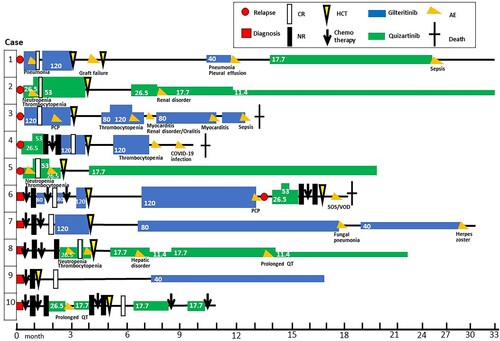
Table 1. Patient characteristics from diagnosis to transplantation.
Table 2. Profile of maintenance therapy.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.