Figures & data
Figure 1. Study design flowchart. Notes: CGMH-KH, Chang Gung Memorial Hospital – Kaohsiung; CMUH, China Medical University Hospital; NCKUH, National Cheng Kung University Hospital; NTUH, National Taiwan University Hospital; RRMM, relapsed/refractory multiple myeloma; TCVGH, Taichung Veterans General Hospital.
Table 1. Patient profiles.
Figure 2. Treatment response (A) Progression free survival (B) Duration of response (Patients ≥ PR). Notes: DoR, duration of response; mPFS, median progression free survival; PR, partial response.
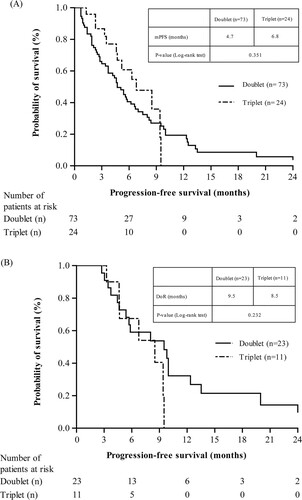
Table 2. Pomalidomide-based treatment response comparison.
Table 3. Factors associated with disease-progression on pomalidomide-based treatment.
Table 4. Healthcare utilization during pomalidomide-based treatment.
