Figures & data
Table 1. Physico-chemical properties of the studied drugs.
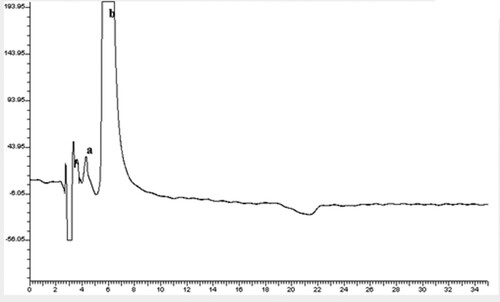
Figure 1. Chiral separation in a chiral medium (Method A) using the described MLC for the separation of racemic mixture of: (a is enantiomer #1, b is enantiomer # 2).
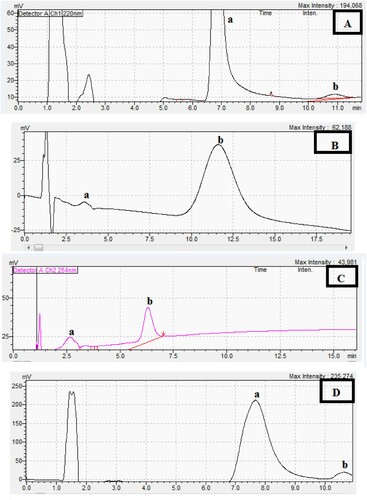
Figure 2. Chiral separation in chiral medium (Method B) using the described HMLC for the separation of racemic mixture of 200.0 µg/mL CTZ on cellulose 1 column: (a is enantiomer #1, b is enantiomer # 2).
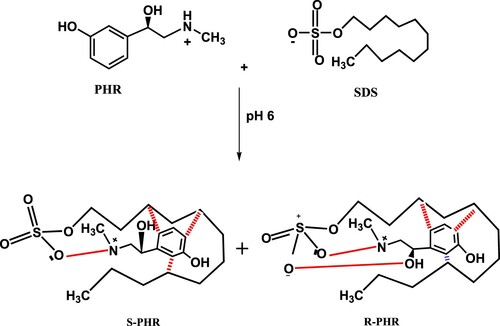
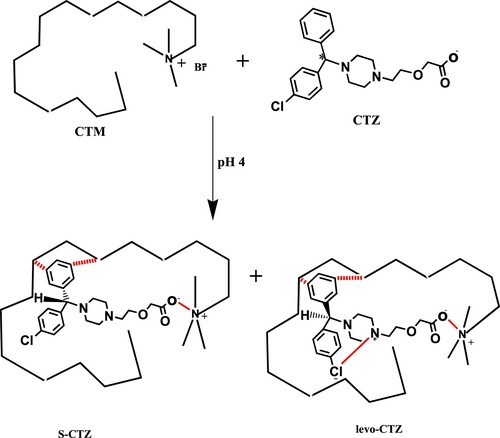
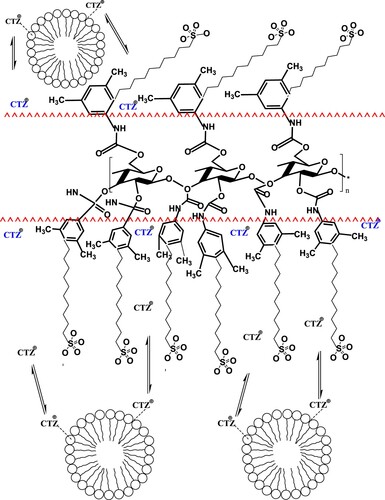
Scheme 1. Postulation of chiral separation mechanism of PHR in achiral medium using the SDS-based mobile phase at pH 6.