Figures & data
Table 1. Characteristics of the covered imaging techniques to study hepatic transporter activity.
Table 2. PET radiotracers to study the activity of hepatic transporters.
Table 3. SPECT and MR imaging agents used to study the activity of hepatic transporters.
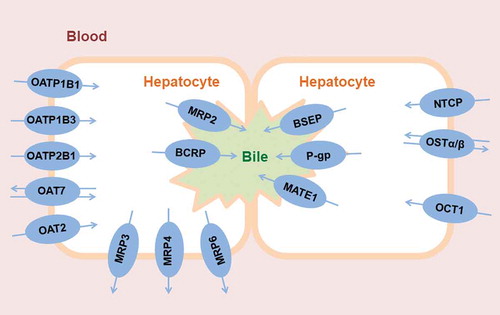
![Figure 2. Pharmacokinetic models developed to study the hepatobiliary disposition of [11C]erlotinib. (a) Four compartment model developed from the combination of two previously developed models [Citation60,Citation89]. This model includes two main regions of interest (ROIs): the liver and the extrahepatic bile duct and gall bladder (eBD/GB). The liver ROI includes compartments representing the amount of radioactivity in the blood fraction in the liver sinusoids used as the model input function (Xblood, 0.25% of the total liver volume), in the hepatocytes (Xhep) and in the intrahepatic bile (Xih, 0.32% of the total liver volume). The extrahepatic ROI compartment represents the amount of radioactivity in the visible part of the extrahepatic bile duct and the gall bladder. The kinetic parameters define the exchange rate of radioactivity between blood and hepatocytes (k1 and k2), from hepatocytes to the intrahepatic bile duct (k3), or from the intrahepatic bile duct to the extrahepatic bile duct and gall bladder (k5). (b) The three compartment model includes compartments representing the blood in the liver sinusoids (used as the model input function), liver tissue (combination of the hepatocytes and the intrahepatic bile duct from the four compartment model) and excreted bile out of the liver ROI. The kinetic parameters define the exchange rate of radioactivity between the blood and the liver (k1 and k2) or from the liver to the excreted bile (k3). The differential equations depicted under the kinetic models are implemented to fit the amount of radiotracer in each compartment to the obtained PET TACs and obtain the estimates of the kinetic parameters. In the equations, X represents the amount of radiotracer in the compartment and ks are the respective rate constants. Note that k1 represents the basolateral uptake rate and does not discriminate between active or passive uptake mechanisms. According to PET pharmacokinetic modeling, the parameter K1 is perfusion-dependent and can be related to blood flow (Q) as: K1 = E x Q; where E represents the unidirectional first-pass extraction ratio [Citation140]. In the present model, k1 can be expressed by means of K1 as k1 = K1 x (Vliver/Vblood), where Vliver corresponds to the volume of hepatocytes in (a) and the volume of liver tissue in (b), and Vblood corresponds to the volume of blood in the hepatic sinusoids. Therefore, the uptake rate constant k1 can be expressed in terms of perfusion as: k1 = (E x Q x Vliver)/Vblood.](/cms/asset/8ad6317e-35d3-430d-95fd-56907003954f/iemt_a_1718107_f0002_oc.jpg)
![Figure 3. Representation of [11C]erlotinib radioactivity concentration in the radial artery (assumed to be equal to the concentration in the hepatic artery), in the mathematically-derived portal vein and in the estimated dual input function in one representative subject. The enlarged graph section shows only the first 10 min of the PET scan duration for which the difference in concentration is larger between arterial and venous hepatic blood. The equations depicted below the graph show the mathematical method implemented to estimate the radiotracer concentration in the portal vein. The concentration in the portal vein along time, CPV(t), can be calculated as the convolution integral between the concentration in the hepatic artery, CHA(t), and the impulse-response function, h(t) as described in the first equation. This impulse-response function (second equation) is mainly characterized by the parameter β which determines the mean transit time for the passage of radiotracer from the intestine to the portal vein. Finally, as indicated in the third equation, the total concentration in the liver sinusoids (dual input function, Cdual(t)) can be represented as a flow-weighted function which takes into account the hepatic artery flow fraction (fHA, ~0.25) and the portal vein flow fraction (fPV, ~0.75).](/cms/asset/01cca2f6-9be2-4b71-9e5e-05348216caf5/iemt_a_1718107_f0003_oc.jpg)
