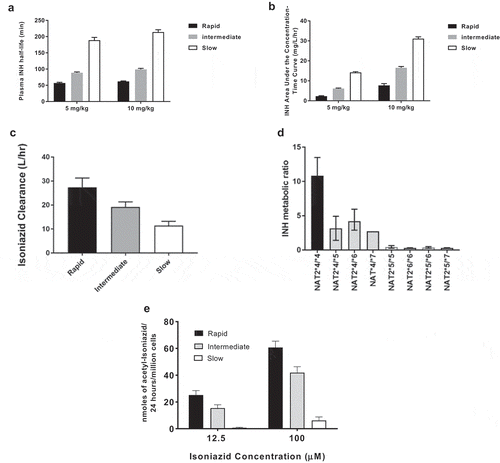
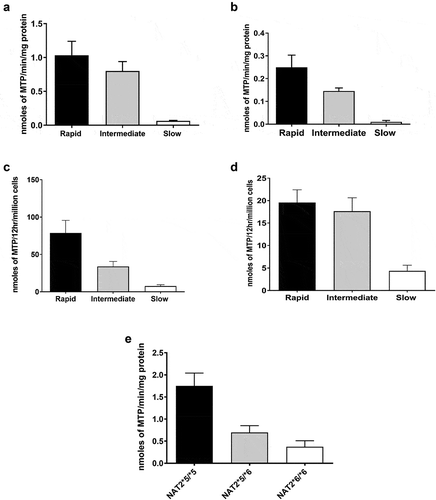
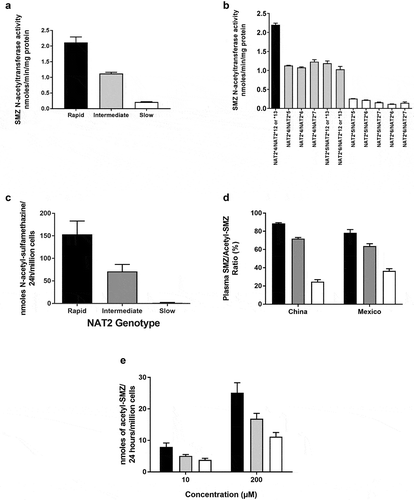
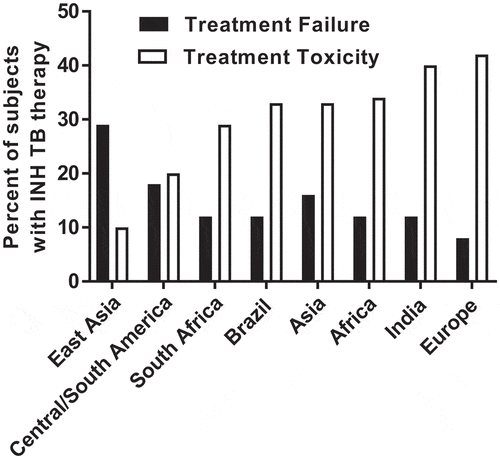
Figures & data
Table 1. NAT2 SNPs and their corresponding alleles/haplotypesa
Table 2. Amifampridine pharmacokinetic parameters in rapid and slow NAT2 acetylators