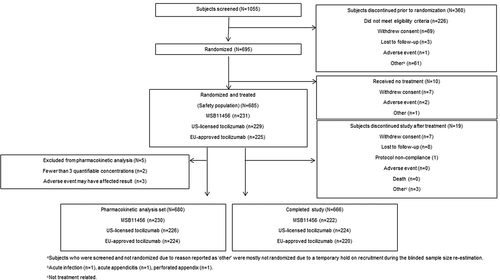
Figures & data
Table 1. Subject demographics and characteristics at baseline, by treatment group and overall (safety analysis seta).
Figure 2. Arithmetic mean (SD) tocilizumab serum concentration–time profiles following a single dose of MSB11456, US-licensed tocilizumab or EU-approved tocilizumab in healthy subjects on a linear scale (pharmacokinetic analysis seta).
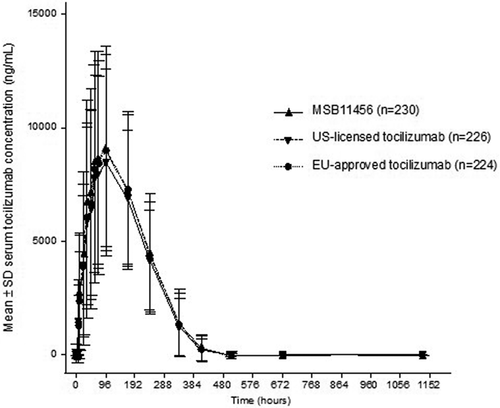
Table 2. Pharmacokinetic results after a single dose of MSB11456, US-licensed tocilizumab or EU-approved tocilizumab in healthy subjects (pharmacokinetic analysis seta).
Table 3. Geometric LS mean ratios for primary pharmacokinetic parameters after a single dose of MSB11455, US-licensed tocilizumab, or EU-approved tocilizumab in healthy subjectsa (pharmacokinetic analysis setb).
Figure 3. Arithmetic mean (SD) sIL-6 R baseline-adjusted serum concentration–time profiles following a single dose of MSB11456, US-licensed tocilizumab or EU-approved tocilizumab in healthy subjects on a linear scale (pharmacodynamic analysis seta).
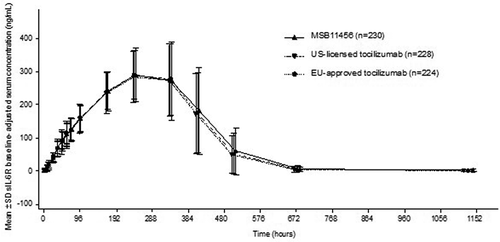
Table 4. Geometric LS mean ratios for pharmacodynamic parameters after a single dose of MSB11455, US-licensed tocilizumab, or EU-approved tocilizumab in healthy subjectsa (pharmacodynamic analysis setb).
Table 5. Safety and tolerability of tocilizumab, by treatment group and overall (safety analysis seta).