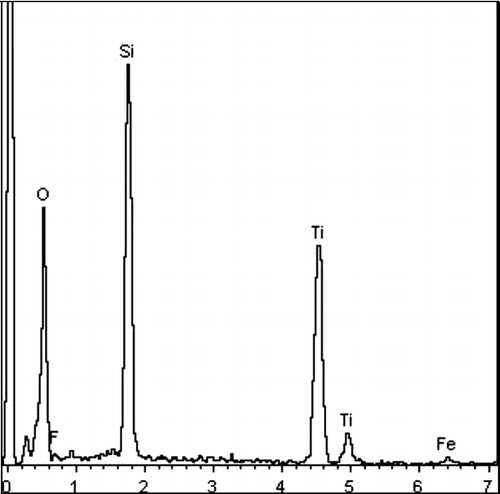
Figures & data
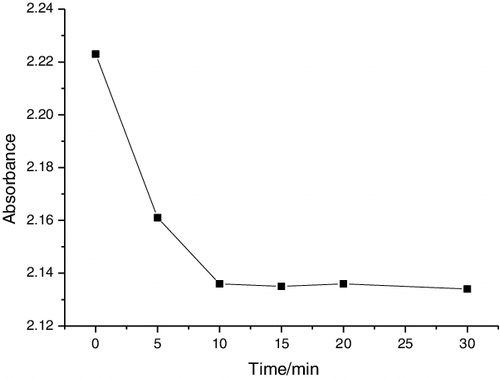
Figure 2. Decomposition kinetics of formaldehyde solutions using pure TiO2, TiO2–SiO2, and Fe3+/F− co-doped TiO2–SiO2 films on glass substrates under visible light irradiation for 200 min.
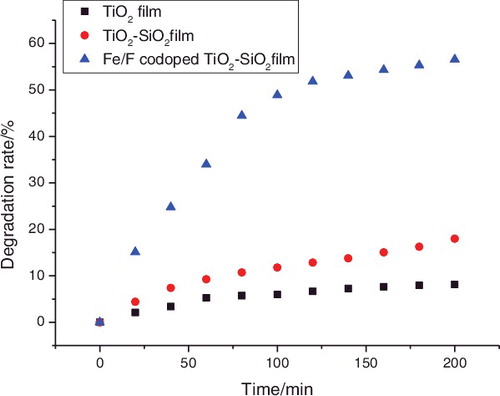
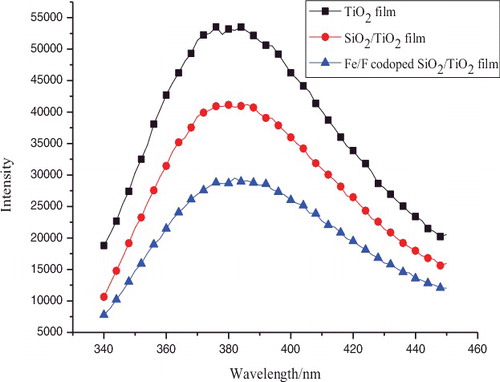
Figure 3. (a) UV–Vis absorption spectra and (b) plots of (αhν)1/2 versus energy (hν) for pure TiO2, TiO2–SiO2, and Fe3+/F− co-doped TiO2–SiO2 films.
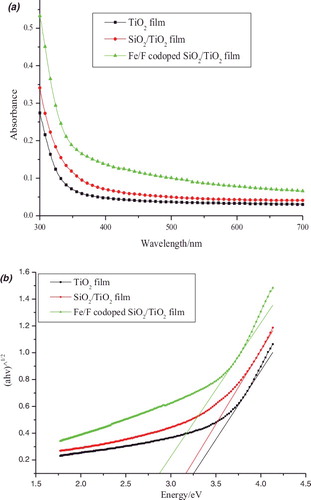
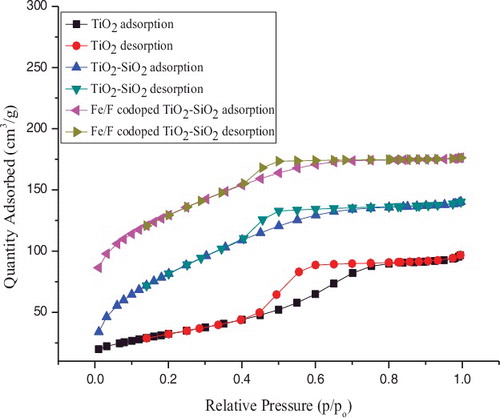
Figure 5. N2 adsorption/ desorption isotherms of pure TiO2, TiO2–SiO2, and Fe3+/F − co-doped TiO2–SiO2 powders.