Figures & data
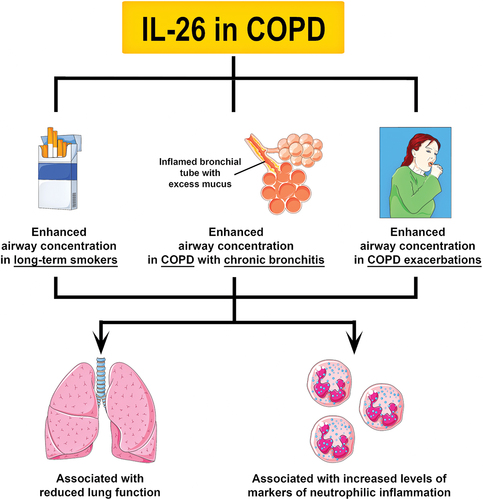
Figure 1. Clinical immunology of IL-26 in the airways. IL-26 is abundantly expressed in human airways. To date, constitutive expression of IL-26 has been reported in both immune and structural cells relevant to human airways. Moreover, the stimuli shown in red can considerably enhance IL-26 expression in these cells. Although IL-26 stimulates the release of pro-inflammatory cytokines (incl. IL-8, TNFα, GM-CSF, and interferons) in alveolar macrophages, monocytes, T cells, and NK cells, it appears to have more complex effects on bronchial epithelial cells and neutrophils. In bronchial epithelial cells, IL-26 stimulation inhibits the release of pro-inflammatory cytokines, but upregulates the IL-26 receptor complex, and its associated transcription factors STAT1 and STAT3. Meanwhile, IL-26 stimulated neutrophils present enhanced chemotaxis, decreased chemokinesis, reduced secretion of myeloperoxidase (MPO) and elastase (NE), and downregulation of the IL-26 receptor complex, STAT1 and STAT3. These varied outcomes suggest that IL-26 induces cell type-specific effects.
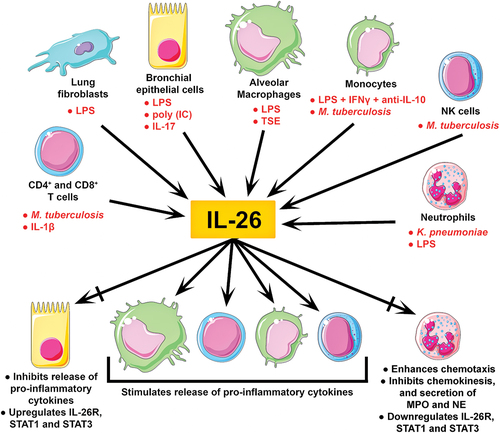
Figure 2. Principal involvement of IL-26 in asthma. One study showed that the concentration of IL-26 in is higher in patients with asthma regardless of disease-severity, degree of control, or atopic status. Furthermore, another study found that patients with severe asthma have increased levels of IL-26 in induced sputum (i.e. proximal airways). In contrast, another study showed that patients with mild-to-moderate asthma have decreased levels of IL-26 in BAL fluid (i.e. distal airways). Thus, alterations in IL-26 expression appear to be compartment-specific and disease severity-dependent in patients with asthma. However, the concentration of IL-26 in induced sputum and BAL fluid is higher in children and adults with uncontrolled asthma, which hints at a potential link between IL-26 and glucocorticoid-resistant phenotypes of asthma. Although it is unclear whether alterations in IL-26 expression could be a causative factor, a consequence, or a defense mechanism of asthma, these changes have been associated with reduced lung function and increased markers of neutrophilic inflammation.
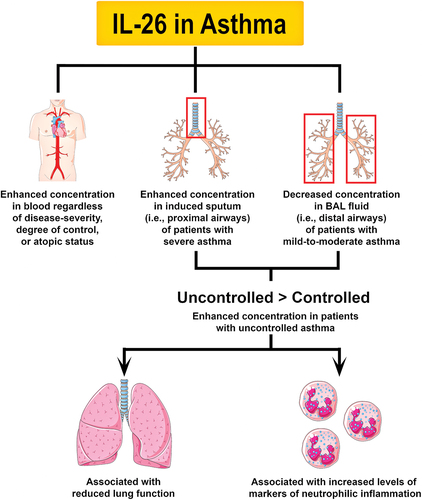
Figure 3. Principal involvement of IL-26 in COPD. To date, two studies have shown that the concentration of IL-26 is increased in induced sputum and BAL fluid from long-term smokers, and COPD patients with chronic bronchitis or exacerbations. Like in asthma, an increase in IL-26 expression has been associated with reduced lung function and increased markers of neutrophilic inflammation in long-term smokers and COPD patients. Notably, an increase in IL-26 in the airways of COPD patients was shown to precede exacerbations. Further research is needed to determine the specific role of IL-26 in COPD.