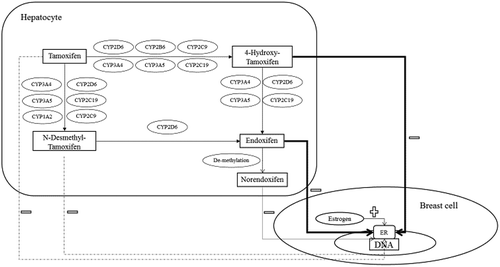
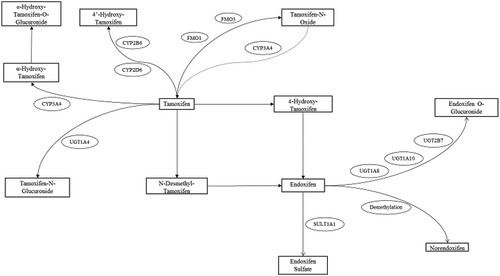
Figures & data
Table 1. Pharmacokinetics parameters of tamoxifen and endoxifen.
Table 2. Minor enzymes involved in tamoxifen metabolism.
Table 3. Minor enzymes involved in endoxifen metabolism.
Table 4. No genetic factors affecting tamoxifen and endoxifen concentrations.