Figures & data
Figure 1. Determining values, μ, for the birth and death rates of invasive Rattus from South Africa.
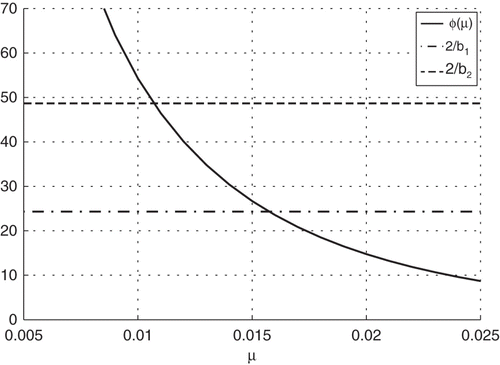
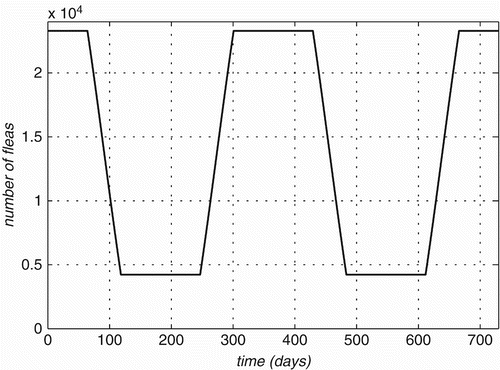
Figure 3. Flea population size variation on a 1000-strong population of each of three Rattus species is plotted over time.
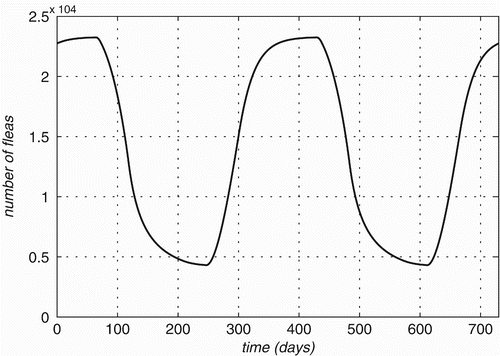
Figure 4. Compartmental structure and epidemiological flow between the compartments of Bartonella infections in Rattus.
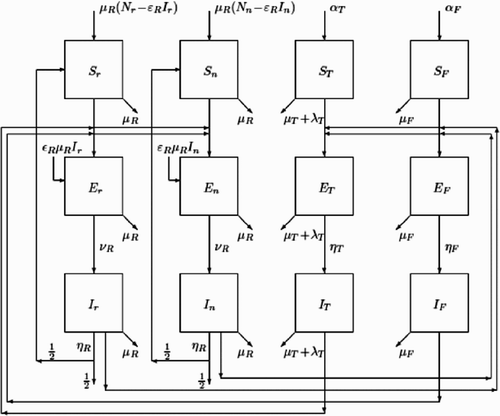
Table 1. Model parameters and baseline values for Bartonella in Rattus.
Figure 5. Changes in compartment sizes over time for Rattus rattus: (a) Case A: in an infection-free environment, the infection is introduced through a single infected individual and (b) Case B: both host and vector populations are initially completely infective.
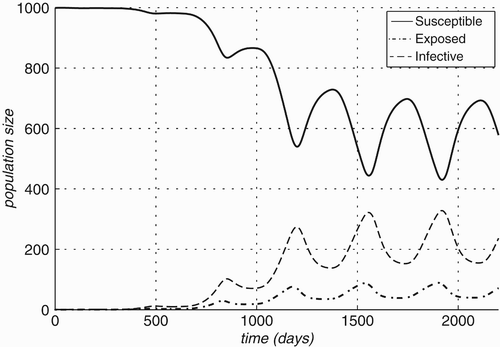
Figure 6. Changes in compartment sizes over time for R. norvegicus: (a) Case A: in an infection-free environment, the infection is introduced through a single infected individual and (b) Case B: both host and vector populations are initially completely infective.
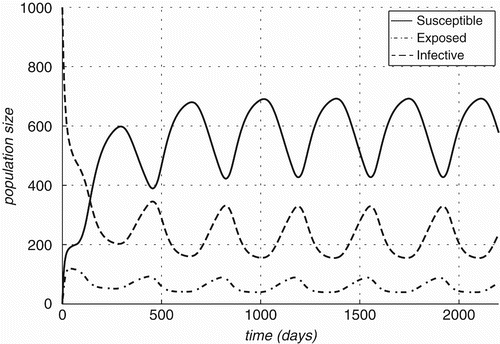
Figure 7. Bartonella infection prevalence rates over time for two host species, R. rattus and R. norvegicus and two vectors, Ixodid ticks and Xenopsylid fleas: (a) Case A: in an infection-free environment, the infection is introduced through a single infected individual and (b) Case B: both host and vector populations are initially completely infective.
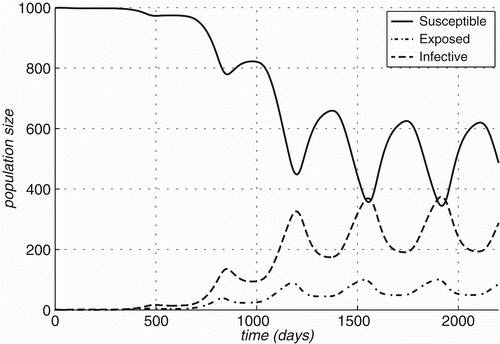
Table 2. Observed average infection prevalence for two Rattus host species and two ectoparasite vectors.
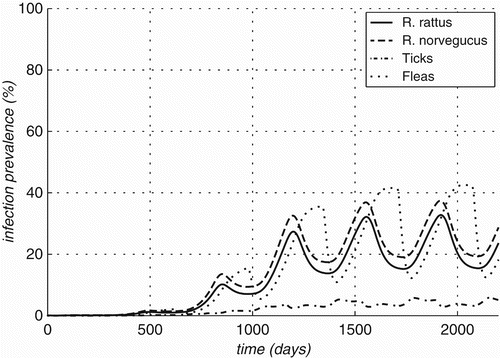
Figure 8. Prevalence rates over time for optimal value of parameters estimated for Bartonella infections in Rattus and two vectors (Ixodid ticks and Xenopsylid fleas).
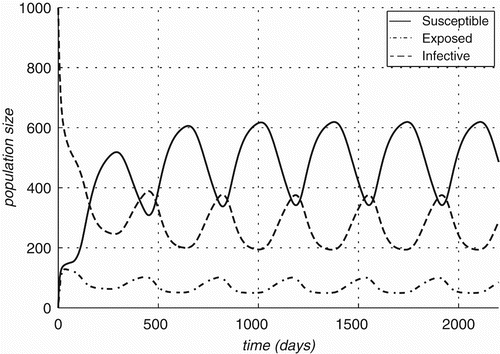
Table 3. Optimal coefficients and model prevalence rates.
Figure 9. Bartonella infection prevalence rates for (a) R. rattus and (b) R. norvegicus and their respective ectoparasite populations as simulated by the mathematical epidemiological model.