Figures & data
Table 1. Synthesis of 5-unsubstituted 3,4-dihydropyrimidinone 1 using 0.1 mmol of different catalysts.
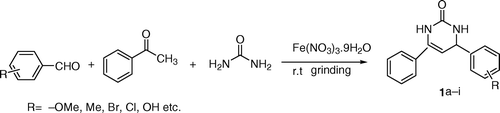
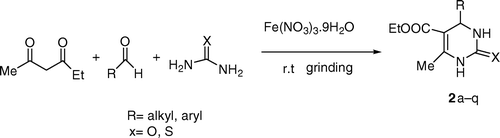
Table 2. Synthesis of 5-unsubstituted 3,4-dihydropyrimidinone 1 and 5-substituted 3,4dihydropyrimidinone 2 derivatives.
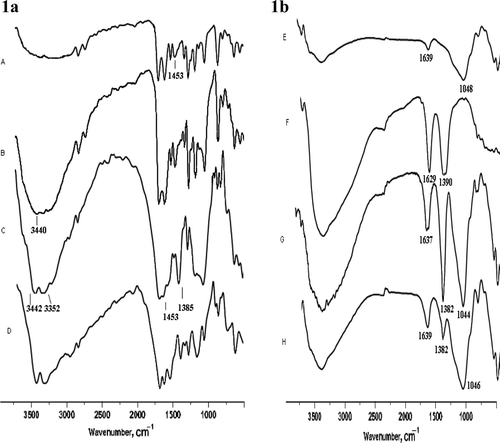
Figure 1. (a) IR spectra of reaction mixture (acetophenone, anisaldehyde, and urea) at various time intervals, 60 mins (A), 120 mins (B), 3 hr (C), and pure product (D) where region 3000–3500 cm−1 represents splitting of νN–H stretching into two peaks and region 1500–1700 cm−1 shows gradual change of νC = O stretching frequencies. (b) IR spectra of Monmorillonite K-10 (E), Fe (NO3)3.9H2O (F), clayfen (G), and regenerated clayfen (H).