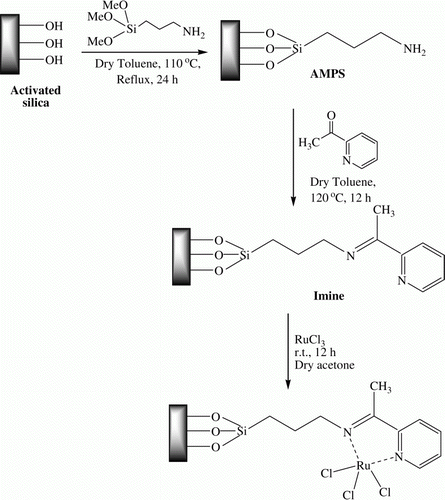
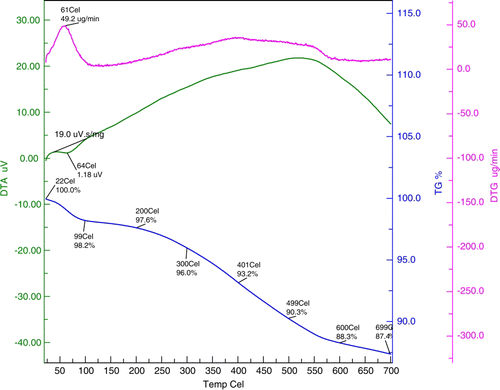
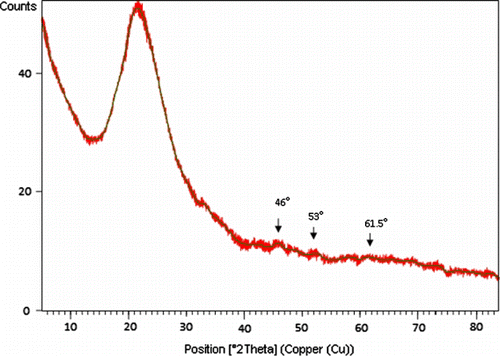
Figures & data
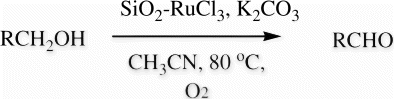
Table 1. Effect of different solvents and catalyst loading for SiO2-RuCl3 catalyzed oxidation of 4-chlorobenzylic alcohol at 80°C.
Figure 5. Recyclability of SiO2-RuCl3. Reaction conditions: 4-chlorobenzylic alcohol (0.143 g, 1 mmol), SiO2-RuCl3 (0.2 g, 3.5 mol% Ru), K2CO3 (2 mmol), acetonitrile (5 mL) at 80 °C for 10 h.
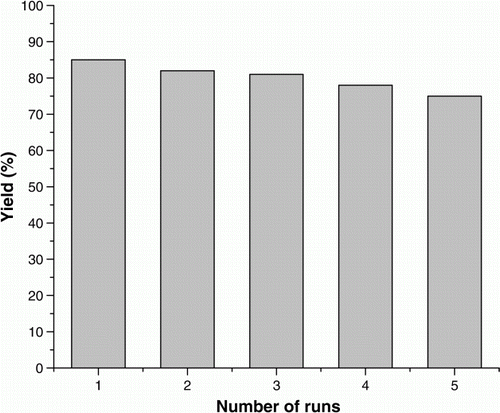
Table 2. SiO2-RuCl3 catalyzed selective oxidationa of benzylic alcohols to corresponding aldehydes by stirring in acetonitrile at 80°C using molecular oxygen as an oxidant.
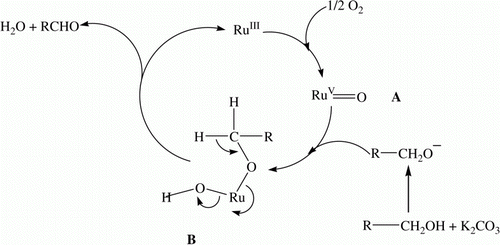
Figure 6. Proposed mechanism for the SiO2-RuCl3 catalyzed selective oxidation of benzylic alcohols to the corresponding aldehydes using molecular oxygen.