Figures & data
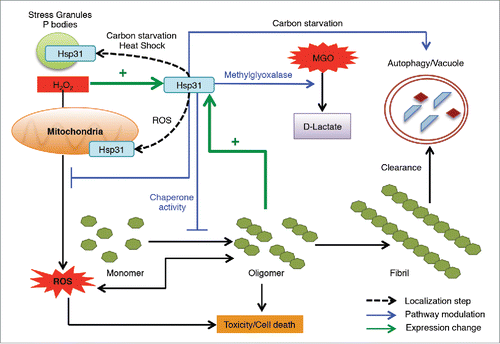
FIGURE 1. The hyperbolic plot of substrate concentration versus rate of D-lactate production by Hsp31 (circles), MORF Hsp31 (squares) and DJ-1 (triangles) are shown and the solid line represents the Michaelis-Menten best-fit model. The Vmax parameters were determined based on this model.
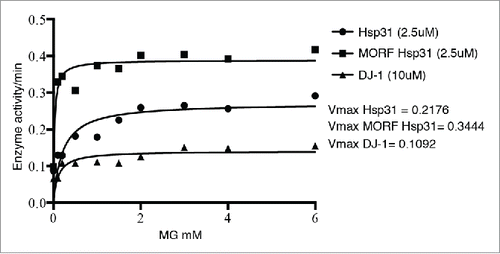
FIGURE 2. Hsp31 cannot resolve the Sup35 amyloids. [PSI+] cells were transferred with pAG415-GPD-Hsp31-DsRed, p2HG Hsp104 and their corresponding empty vectors. Cultures were incubated at 30°C for 16 hrs before plating on ¼ YPD medium to develop colony color. Change from white to red color phenotype demonstrated curing. Hsp104 is able to cure the [PSI+] phenotype as seen by the red colonies. Hsp31 expression under these conditions can transiently reduce prion aggregation as shown by semi-denaturing gels and microscopy but is not able to cure prions.
![FIGURE 2. Hsp31 cannot resolve the Sup35 amyloids. [PSI+] cells were transferred with pAG415-GPD-Hsp31-DsRed, p2HG Hsp104 and their corresponding empty vectors. Cultures were incubated at 30°C for 16 hrs before plating on ¼ YPD medium to develop colony color. Change from white to red color phenotype demonstrated curing. Hsp104 is able to cure the [PSI+] phenotype as seen by the red colonies. Hsp31 expression under these conditions can transiently reduce prion aggregation as shown by semi-denaturing gels and microscopy but is not able to cure prions.](/cms/asset/4a753cd8-d583-4628-9413-62713196919f/kprn_a_1141858_f0002_oc.gif)
TABLE 1. Functional summary of Hsp31 and paralogs.
FIGURE 3. The homeostatic functions of Hsp31 associated with protecting cells from stress. Hsp31 is a methylglyoxalase that converts MGO into D-lactate independent of glutathione. Proteotoxic stress induced the expression of Hsp31, which exerts a protective function against toxic effect of oligomers in yeast cells. Oxidative stress induces the expression of Hsp31, re-localizes it to mitochondria resulting in reduced levels of ROS. Response to other stresses leads to Hsp31 localization to P bodies and stress granules. HSP31 deletion under carbon starvation compromises the autophagy pathway, which is a pathway used to clear oligomerized or aggregated proteins. Despite the role of Hsp31 in autophagy, it has a protective effect against α-Syn oligomerization independent of its role in autophagy because of its inhibitory effect early in the oligomerization process.