Figures & data
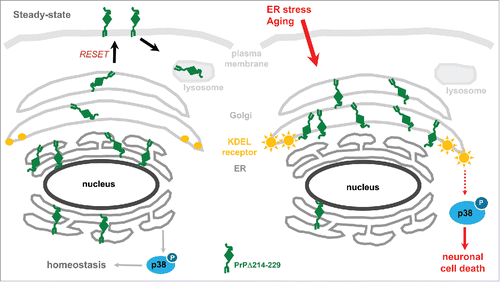
FIGURE 1. Schematic representation illustrating mechanisms of the neurotoxic effect of PrPΔ214-229. (A) In steady-state conditions, PrPΔ214-229 is found mainly at the ER but also slightly at the Golgi and plasma membrane probably following the RESET pathway. In our cell model (but not in the transgenic mice) we see activation of p38-MAPK at steady state levels. We hypothesize that, in this case, this activation leads to protein homeostasis. (B) After applying external stress conditions to cells or, in the case of Tg(PrPΔ214-229) mice due to aging, PrPΔ214-229 leaves the ER in order to follow the RESET pathway for degradation but, due to the mutation, is retained at the Golgi. We hypothesize that this leads to activation of Golgi-resident receptors (e.g., KDEL receptor) leading to increased p38-MAPK phosphorylation and cell death.