Figures & data
Figure 1. The amino acid sequences of the heavy chain variable region of murine 37E1B5. The CDR regions are shown in italic and underlined. The N-linked glycosylation signals, NLS and NYT at the positions 19-21 and 59-61, are high-lighted as yellow, respectively.
Figure 2. Characterization of Beta8 glycosylation. Relative quantitation of released glycans (Fc and Fab) by 2-aminobenzamide (2-AB)-labeled oligosaccharide profiling (A). Site-specific glycan analysis by reduced antibody LC-MS of heavy chain combined, de-convoluted mass spectra for Beta8 (B) and the aglycosylated Beta8-AG (N59Q) variant (C).
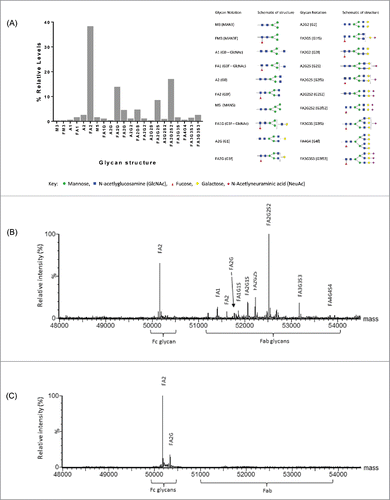
Figure 3. Binding of Beta8 and Beta8-AG to αvβ8 (A). Recombinant αvβ8 was coated to the ELISA wells. Antibody binding was detected using a HRP-conjugated goat anti-human Fc antibody. Inhibition of αvβ8 binding to LAP ((B) and C). Recombinant LAP was coated to the ELISA wells. The amount of the recombinant αvβ8 protein that generated 75% maximum binding signals was mixed with serially diluted Beta8 and Beta8-AG (B) or Fabs of Beta8 and Beta8-AG (C), and the reaction was incubated at room temperature. The ability of αvβ8 binding was detected by an HRP-conjugated anti-αvβ8 antibody.
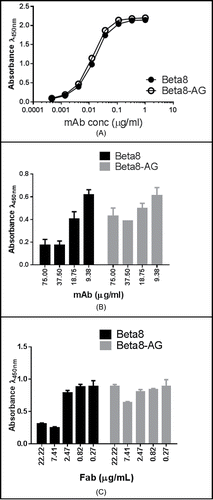
Figure 4. Inhibition of TGF-β activation by 37E1B5 requires a complex glycan. Schematic overview of the different antibody glycoforms assessed for bioactivity and PK properties, illustrated for the binantennary glycan FA2G2S2 (A). TMLC-HeLa cell co-culture TGF-β signaling reporter assay. Relative luciferase units (RLU) from TMLC reporter cells show baseline luciferase reporter activity (“TMLC”). Relative luciferase units (RLU) from TMLC and HeLa αvβ8+ cell co-culture show maximal luciferase reporter activity (“TMLC + HeLa); and the anti-TGF-β mAb 1D11 (“anti-TGFb-1D11,” 7.5 µg/mL) effect on the TGF-β dependent luciferase reporter activity in the TMLC and HeLa αvβ8+ co-culture. Mouse IgG1 (“mIgG1,” 7.5 µg/mL) was used as the isotype control. (B). Maximum response of TGF-β dependent luciferase reporter activity by 37E1B5 variants in the TMLC co-culture assay with HeLa αvβ8+ cells. (C). Data shown is the mean ±SEM from 3 independent experiments.
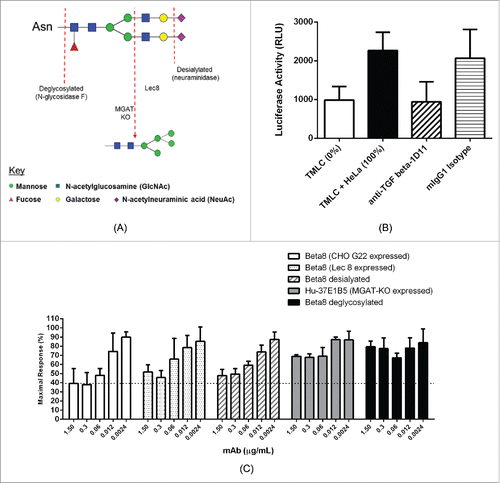
Table 1. Model-based pharmacokinetic parameter estimates for comparison of the wild type (WT), fully glycosylated (AG) and the desialylated (DE) Beta8 in mice. PK parameters derived from the non-linear mixed effects software NONMEM (Icon Development Solutions, Version 7.3).
