Figures & data
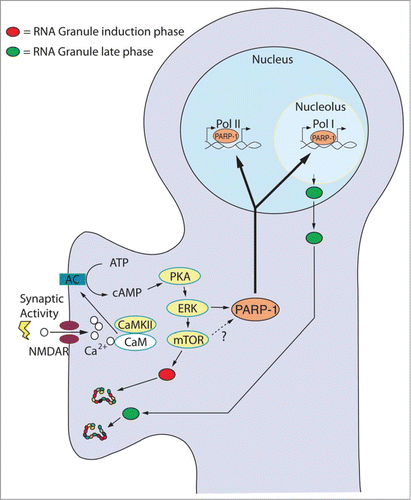
Figure 1. Hypothetical Model for the Transduction of Synaptic Stimuli to Long-Term Plasticity. Synaptic stimulation triggers adenylate cyclase (AC) resulting in the rapid release of cAMP and the activation of the cAMP-PKA-ERK pathway. Stimuli leading to long-term plasticity activate mTOR-dependent translation of preexisting RNA granules (red). Simultaneously, the PKA-ERK pathway induces the synthesis and activation of chromatin remodeling factors (e.g. PARP-1) that opens the chromatin allowing plasticity-dependent transcription to take place. Crucial among the new transcripts are precursor rRNAs required for the formation of new ribosomes. We hypothesize that new and qualitatively different ribosomes are assembled into new RNA granules (green) and shipped to activated synapses to maintain, through local protein synthesis, the long-lasting changes required for long-term synaptic plasticity and memory.