Figures & data
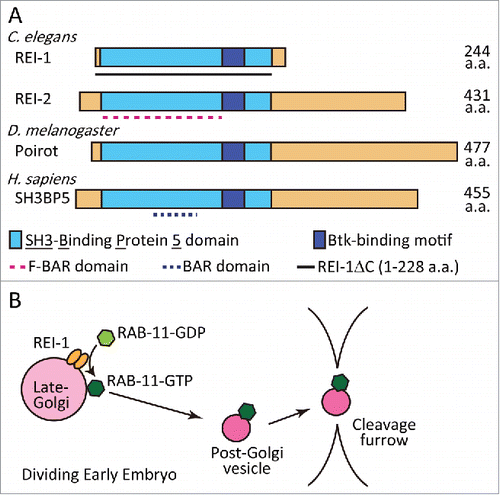
Figure 1. (A) Domain structure of the REI-1 family of proteins. Our results show that almost full-length REI-1 (1-228 aa) is required for RAB-11.1 binding. (B) A model describing the role of REI-1 in RAB-11 activation and translocation. REI-1 activates RAB-11, by exchange of GDP with GTP, principally at late-Golgi membranes. RAB-11-positive post-Golgi vesicles or compartments are then targeted to the cell cortex and cleavage furrow to regulate cytokinesis and membrane traffic.