Figures & data
Figure 1. High-fat feeding reduces aspects of gut immunity in mice. Quantification of neutrophil myeloperoxidase (MPO) levels in small intestine, ileum, and colon gut segments of WT (A) and NOD2-null (B) 16-week chow- and HFD-fed mice. For all groups, n = 5. * indicates P <0.05.
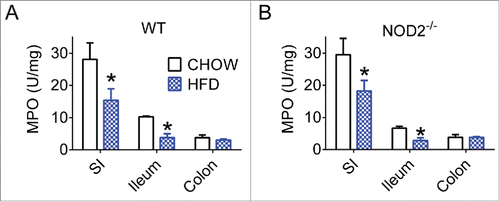
Figure 2. High-fat diet impairs gut Th17 immune responses. Quantification of various Th1, Th2, and Th17-related cytokines in small intestine, ileum, and colon gut segments of WT (A, E, I) and NOD2-null (B, F, J) 16-week chow- and HFD-fed mice. Host genotype-specific effects were observed during dietary stress for IL-12p40 in small intestine (C, D) and IL-17A in ileum (G, H). Data is presented as pg cytokine per mg of indicated tissue. For all groups, n = 5. * indicates P <0.05.
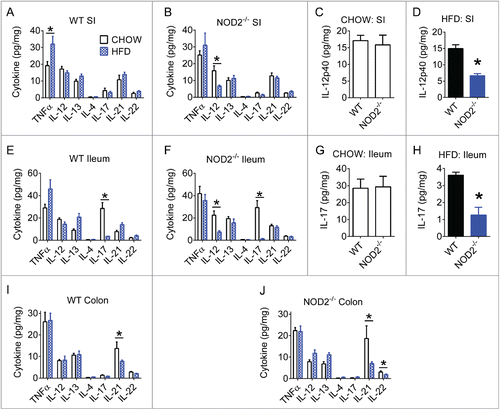
Figure 3. High-fat diet has different effects on Th17 immunity in metabolic tissues. Quantification of various Th17 cytokines in white adipose (A, B) and liver (C, D) tissue of WT and NOD2-null chow- and HFD-fed mice. Data is presented as pg cytokine per mg of indicated tissue. For chow-fed animals, n = 10. For HFD-fed animals, n = 11 (WT) or n = 8 (NOD2-null). * indicates P <0.05.
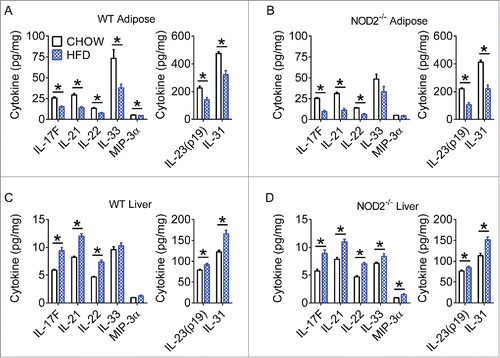
Figure 4. Diet, host genotype, and microbes link compartmentalized Th17 immunity. A proposed model indicating how dietary factors, host genetics, and microbiota connect gut immunity to metabolic tissue inflammation. An obesity-promoting diet alters the balance between gut immunity and microbiota composition, and defective NOD2 worsens these effects. This environment is characterized by decreased Th17 immune responses that contribute to bacterial cell and component evasion of the gut mucosal barrier and dissemination into host circulation. Destinations for gut bacterial products include tissues such as adipose and liver, which in turn experience increased metabolic inflammation and subsequent impaired insulin sensitivity.