Figures & data
Figure 1. NleA is modified by O-linked glycosylation. Western blot analysis of immunoprecipitate from CHO ldlD cells infected with EPEC UMD207 NleA-FLAG probed with vicia villosa lectin (VVL), and anti-FLAG, and anti-NleA antibodies. Cells were cultured in media without (-), with galactose (G), with GalNAc (GN), or with both galactose and GalNAc (+) sugar supplementation. Migration of molecular weight markers (kDa) is indicated on the left.
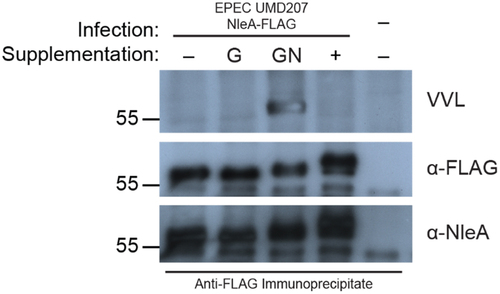
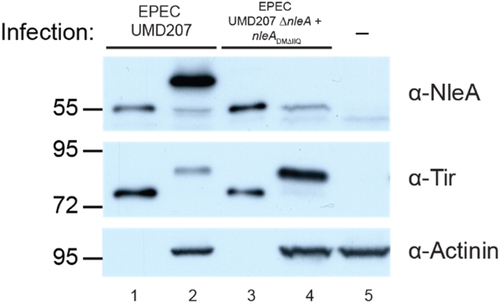
Figure 2. NleA deficient in Sec24 binding does not get modified. Western blot analysis of HeLa cell lysate infected with EPEC UMD207 expressing WT NleA (lane 2), NleADMΔIIQ (lane 4), or uninfected (lane 5). Bacterial lysates from the indicated strain are present in lanes 1 and 3. Blots were probed with anti-NleA, anti-tir, and anti-actinin antibodies. Migration of molecular weight makers (kDa) is indicated on the left.
Data availability statement
The data that support the findings of this study are available from the corresponding author, SG, upon reasonable request.
