Figures & data
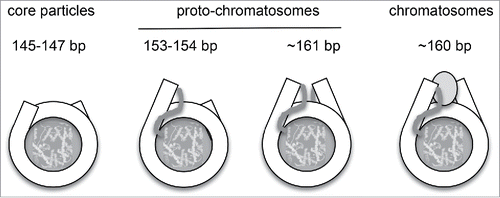
Figure 1. Simultaneous digestion of chromatin with MNase and ExoIII reveals new intermediate mono-nucleosomal particles. DNA length distributions obtained by paired-end sequencing of nucleosomes from yeast cells. (A) MNase only. (B) MNase and ExoIII. Adapted from Fig. 1 in Citation1.
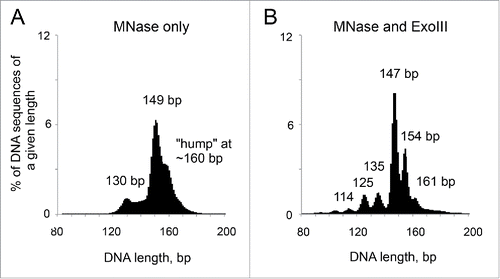
Figure 2. Digestion of chromain by MNase and ExoIII. Adapted from Fig. 1 in Citation1.
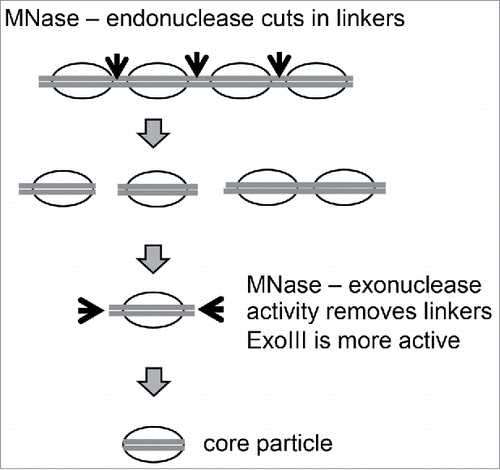
Figure 3. Structural relationship between nucleosome core particles, proto-chromatosomes and chromatosomes. Views from above, drawn approximately to scale, based on the nucleosome structure. Citation2 The final 10 bp on each side of the nucleosome core are almost straight, projecting a short distance out of the particle. Proto-chromatosomes are shown with an extra 7 bp on one side (154 bp) or both sides (161 bp) with a continuing straight trajectory. We propose that the fundamental particle is the 161-bp proto-chromatosome, which has both extensions. We suggest that each extension is protected by a specific but unidentified core histone-DNA contact, most likely a histone tail (as indicated), to form the H1 binding site. The chromatosome is formed when H1 binds (shown here as a light gray oval in a symmetrical location although this is controversial). Adapted from Fig. 6 in Citation1.