Figures & data
Figure 1. Spinal cord degeneration in a mouse model of ADLD. (A) PLP-FLAG-LMNB1 transgenic mice (TG), with oligodendrocyte specific overexpression of lamin B1, show age dependent degenerative phenotypes including kyphosis (black arrow), forelimb paralysis (white arrowhead) and muscle wasting at 13 months while wild type (WT) littermates show no obvious phenotypes. (B) Cervical spinal cords section of TG mice show significant vacuolar degeneration involving the white matter (arrows). No such alterations are observed in spinal cord sections from WT littermates. Top panel – H&E staining, Bottom panel – Fluormyelin staining (Fluromyelin is a fluorescent dye that specifically stains white matter). Reproduced with permission from Rolyan et al., (2015).Citation19
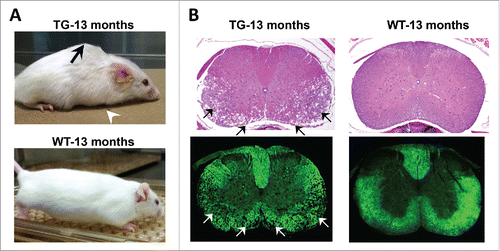
Figure 2. ADLD disease mechanism. Potential disease mechanisms underlying ADLD. Results from our group suggest that lipid dysregulation mediated by age dependent epigenetic alterations in oligodendrocytes are a major driver for the demyelination observed in an ADLD mouse model. However, other pathways such nuclear structural defects or aberrant splicing may also contribute to oligodendrocyte dysfunction. Findings that elevated reactive oxygen species (ROS) promote an accumulation of lamin B1 may provide an alternative hypothesis to explain the late age onset of the disease phenotype.
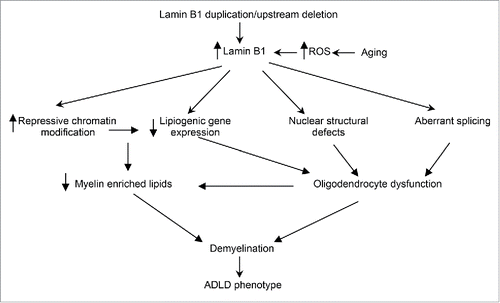
Table 1. Lipid synthesis genes downregulated in 3 and 13 month transgenic PLP-FLAG-LMNB1 mice.
