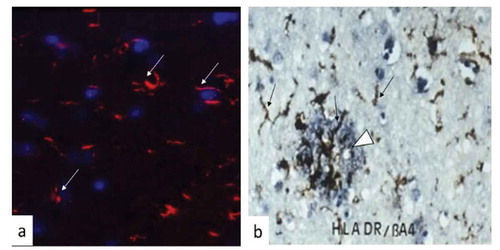
Figures & data
Table 1. Potential mechanisms for microglia affection by Porphyromonas gingivalis (P.g.).