Figures & data
Figure 1. Patient flow diagram. aPatients who were excluded due to adherence criteria, and who died prior to the end of the follow-up period are listed in both of the exclusion categories; bPatients who were excluded, were excluded due to lack of adherence. To be included, all patients had to be on BF Spiromax for the entire follow-up, with a minimum of two prescriptions. BF: budesonide+formoterol; COPD: chronic obstructive pulmonary disease.
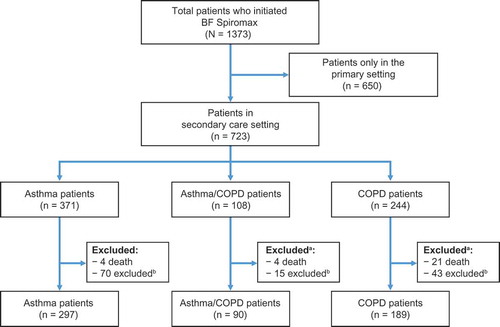
Table 1. Patient characteristics among an eligible patient population.
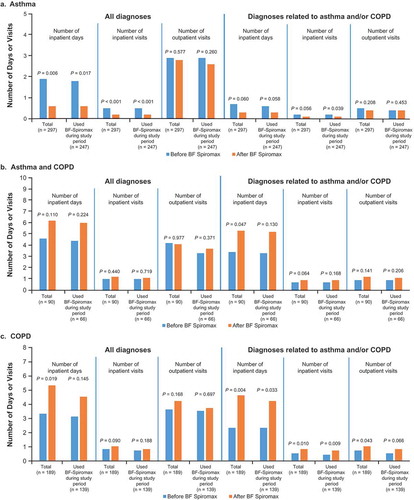
Figure 2. Percentage of patients with an exacerbation before and after initiating BF Spiromax for the asthma patient group (a), the asthma/COPD patient group (b), and the COPD patient group (c). BF: budesonide+formoterol; COPD: chronic obstructive pulmonary disease; ICS: inhaled corticosteroid; LABA: long-acting β2-agonist.
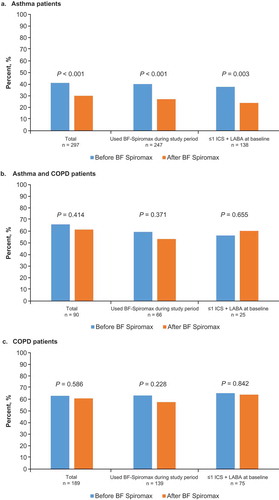
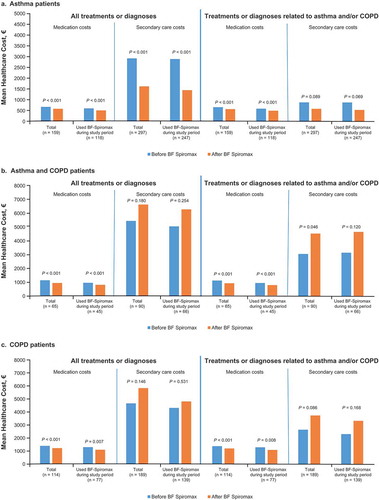
Table 2. Asthma/COPD treatments before and after initiation to BF Spiromax in the total population.