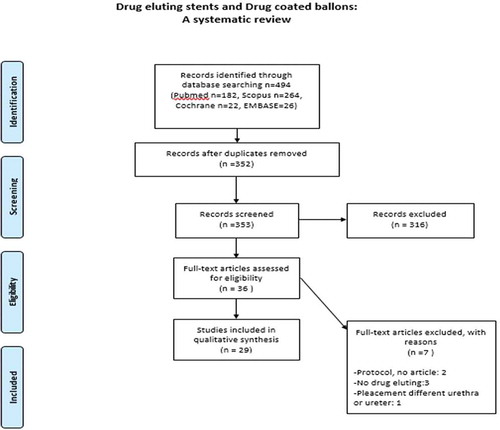
Figures & data
Table 1. Eligibility criteria of the systematic review
Table 2. Comparison of the experimental (in vitro) trials of DES/DCS
Table 3. Comparison of the clinical (in vivo) trials of DES/DCS
Table 4. Comparison of the clinical trials (in vivo) of DCBs