Figures & data
Table 1. Study selection criteria to identify trials for the SLR.
Table 2. List of publications and key trial characteristics, arranged by trial.
Table 3. List of publications and key patient characteristics, arranged by trial.
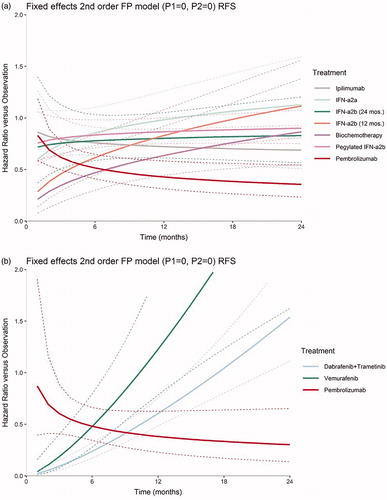
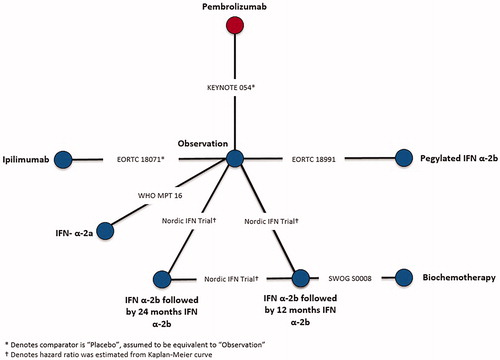
Table 4. Time-varying hazard ratios of recurrence-free survival at select follow-up times for competing interventions vs observation, Stage III.
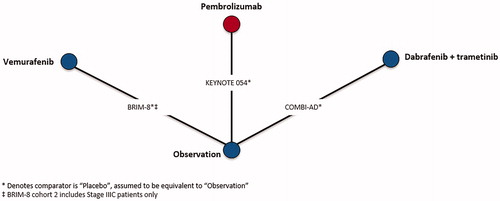
Table 5. Time-varying hazard ratios of recurrence-free survival at select follow-up times for competing interventions versus observation, BRAF + sub-group analysis.