Figures & data
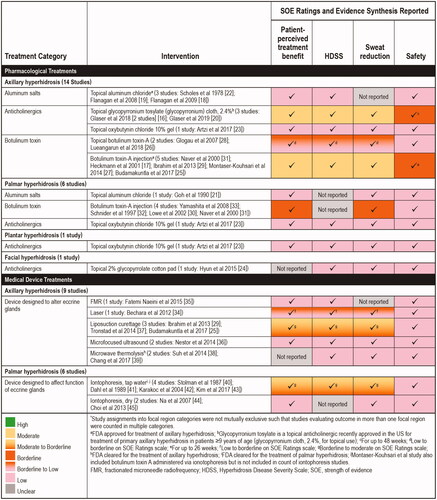
Table 1. Summary and risk of bias for aluminum salt studies.
Table 2. Summary and risk of bias for anticholinergic agents.
Table 3. Summary and risk of bias for botulinum toxin type A studies.
Table 4. Summary and risk of bias for medical devices that alter eccrine glands.
Table 5. Summary and risk of bias for iontophoresis studies.