Figures & data
Fig. 1. POM images. (Top) CLC electrolyte solution containing HMSBT as a monomer (before polymerisation). (Bottom) CLC electrolyte solution after the electrochemical polymerisation.

Table 1 Chemical structures of the monomers, the nematic LC, the chiral inducer, and the supporting electrolyte.
Fig. 2. POM images. (Top) CLC electrolyte solution containing BET as a monomer (before polymerisation). (Bottom) CLC electrolyte solution after the electrochemical polymerisation.

Fig. 3. POM image of the polymer film prepared in the CLC electrolyte solution. (Top) poly HMSBT, (bottom) poly BET.

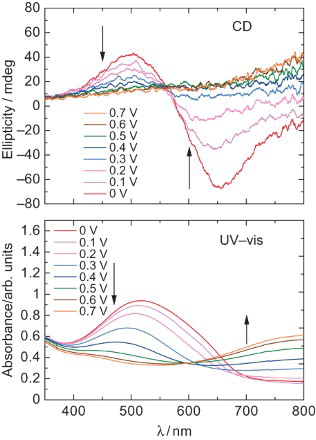
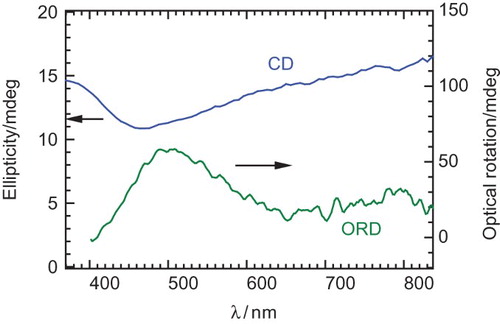
Fig. 4. (Top) CD spectra of the polymer at 0–0.7 V. (Bottom) UV–VIS absorption spectra of poly HMSBT at 0–0.7 V.