Figures & data
Figure 1. Combined chemoimmunotherapy of TRAMP mice with docetaxel and DC-based vaccines. (A) Approximately 8-week-old male TRAMP mice were treated with DTX alone (30 mg/kg), pulsed or unpulsed DC-based vaccines (2 × 106 cells/mouse), or with their combination. (B) Dotplots of GUT expressed as weight of genitourinary tract (GUT) per mouse body weight. *P<0.05 vs. control (t-test).
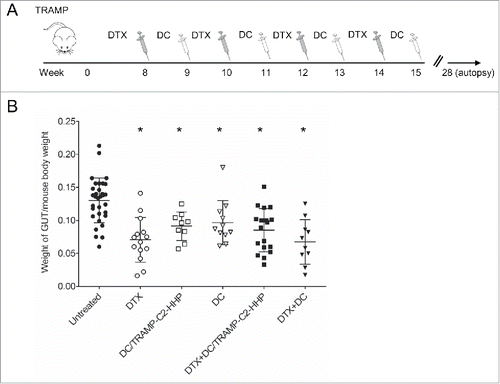
Figure 2. Histopathology analysis of TRAMP mice treated with docetaxel and DC-based vaccines. (A) Quantitative analysis of therapeutic response scoring in TRAMP mice treated with docetaxel and DC-based vaccines. (B) Representative pictures of scoring. (C) Ki67 labeling index and representative pictures of all treated groups and untreated control (D). *P<0.05 vs. control (t-test).
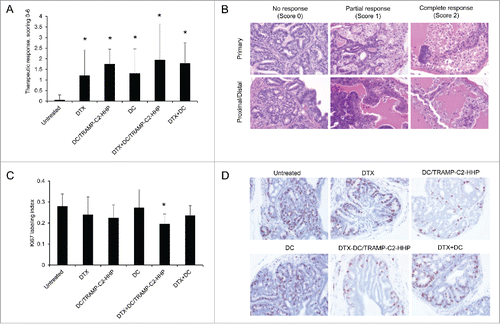
Figure 3. Combined chemoimmunotherapy of TRAMP mice with CY and DC-based vaccines. (A) Approximately 8-week-old male TRAMP mice were treated with CY alone (200 mg/kg), pulsed DC-based vaccine (2 × 106 cells/mouse), or with their combination. (B) Dotplots of GUT expressed as weight of genitourinary tract (GUT) per mouse body weight. (C) Quantitative analysis of therapeutic response scoring in TRAMP mice treated with CY and DC-based vaccines. *P<0.05 vs. control (t-test).
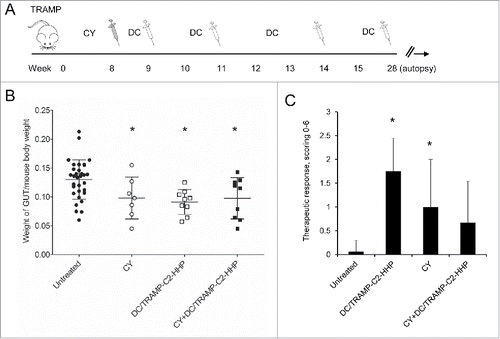
Figure 4. Immmunotherapy of surgical minimal residual tumor disease of TC-1 and TRAMP-C2 tumors with DC-based vaccine. (A) Mice were inoculated s.c. with TC-1 (5 × 104 cells) or TRAMP-C2 (106 cells). When the transplanted tumors reached ∼5–10 mm in diameter, the tumors were excised. DC-based vaccines (2 × 106 cells/mouse) were administered on days 7 and 21 after surgery (left panel). In the case of pretreatment, DC-based vaccines (2 × 106 cells/mouse) were additionally administered on days -7 (TC-1, TRAMP-C2) and -21 (TRAMP-C2) before surgery (right panel) (B) Growth of TRAMP-C2 tumor recurrences after treatment with unpulsed and pulsed DC-based vaccine (left panel) and the growth of TRAMP-C2 tumor recurrences after pre-treatment or treatment with pulsed DC-based vaccine (right panel). (C) Growth of TC-1 tumor recurrences after treatment with unpulsed and pulsed DC-based vaccine (left panel) and the growth of TC-1 tumor recurrences after pre-treatment or treatment with pulsed DC-based vaccine (right panel). *P<0.05 vs. control (Analysis of Variance).
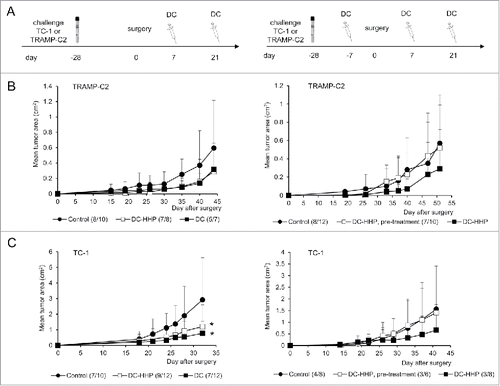
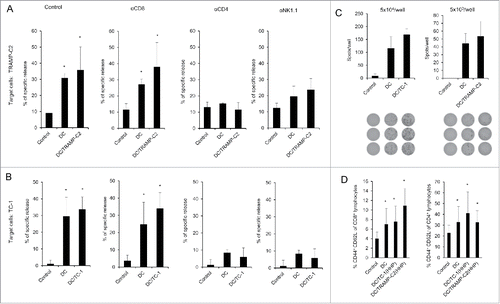
Figure 5. Analysis of splenocytes from mice immunized with pulsed and unpulsed DC-based vaccine. Mice were immunized 2 times in a 2-week interval with 2 × 106 DC pulsed with HHP-treated TC-1 or TRAMP-C2 tumor cells. Ten days after the last immunization, pooled splenocytes of 3 mice were used for in vitro analysis. (A) 51Cr microcytotoxicity assay of depleted splenocytes (αCD8, αCD4, αNK1.1) from mice immunized with DC-based vaccines unpulsed or pulsed with HHP-treated TRAMP-C2 tumor cells or (B) TC-1 tumor cells. (C) The number of IFNγ-producing cells (ELISPOT assay). (D) Percentage of CD44+ CD62L− of CD8+ lymphocytes or CD4+ lymphocytes in splenocytes from mice immunized with pulsed or unpulsed DC-based vaccines. *P<0.05 vs. control. Statistical significances were determined by Student's t-test. Results are representative of 2 independent experiments.