Figures & data
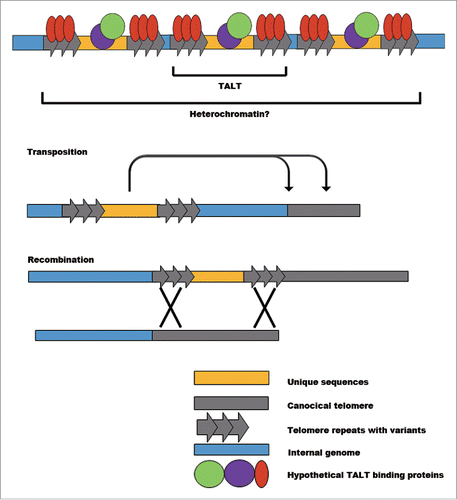
Figure 1. Telomerase-independent telomere maintenance mechanisms. After telomerase loss, alternative mechanisms maintain telomere length. In cancer cells, variant telomere repeats are interspersed within telomeres after recombination-dependent lengthening. Retrotransposon is adopted in many organisms. An example is Drosophila. In Allium cepa, telomeres are protected by other genomic sequences such as minisatellite and rDNA.
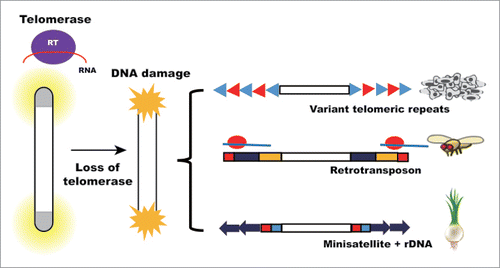
Figure 2. Possible mechanisms of TALT duplication in telomere maintenance. Each TALT has its own unique sequence flanked by telomere repeats. While the protein products encoded by unique sequences do not seem to be critical players in regulating telomere maintenance, specific DNA binding proteins may have functional roles such as inducing heterochromatin, upregulating transposition activity and enhancing recombination events.