Figures & data
Table 1. Baseline characteristics
Figure 1. Hemagglutination inhibition antibody responses before (day 1) and after (day 22) influenza vaccination, and cellular proliferation before (day 1) and after (day 8) influenza vaccination. Both immune responses are presented for the 3 influenza strains of the vaccine (H3N2, H1N1, and B), in fatigued (A and C) and non-fatigued (B and D) cancer survivors. Antibody titres are presented as absolute numbers and mean on a linear scale, the dotted line indicates the protective titer cut-off value. Proliferation counts are presented as absolute numbers and mean (horizontal lines) on a logarithmic scale.
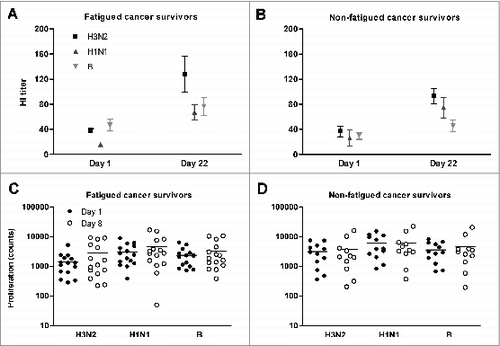
Table 2. Humoral and cellular immune responses
