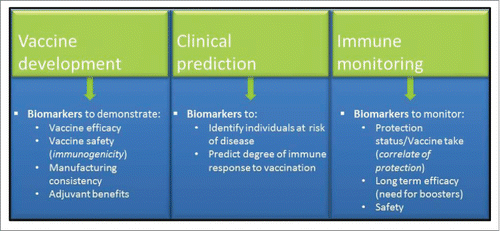
Figures & data
Table 1. Participant list for the Next Generation Vaccine Biomarker Workshop. Contributors represented government, industry, regulatory bodies, academia, public-private partnerships, and funding agencies
Figure 2. CIRN Infrastructure. CIRN is comprised of 5 core networks, as well as 3 support networks, that work collaboratively to advance vaccine research in Canada.
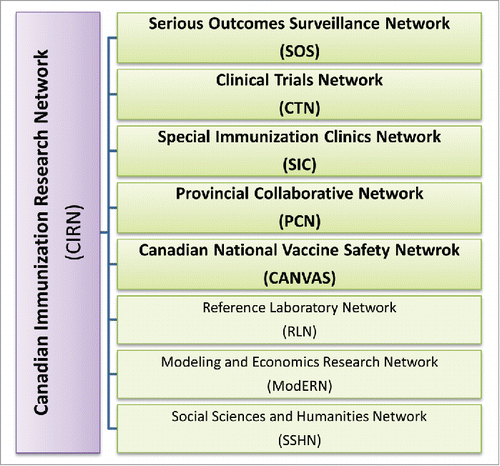
Table 2. Comparison of biomarkers for prophylactic and therapeutic vaccines. For prophylactic vaccines, it is often not possible to monitor efficacy directly (post exposure or challenge), and so a surrogate may be established to demonstrate efficacy. In contrast, for therapeutic vaccines, surrogates are not used for regulatory approval since it is necessary to show direct efficacy in the disease state
Table 3. US FDA Centers and Regulatory Divisions of labor. These centers are involved in both biologics and vaccines approval
Table 4. National Research Council Canada Industrial Research Assistance Program services. The services offered by this program support technological development for Canadian SMEs. Their suite of services, including funding and advisors, aim to help translate ideas into commercial success