Figures & data
Table 1. Characteristics of study participants.
Figure 2. Participant flow chart.
ATP, according-to-protocol; N, number of participants; HBc, hepatitis B core antigen.Note: *One participant also had positive anti-HBc status 1 month post-challenge dose.

Table 2. Seropositivity/seroprotection rates and antibody GMCs, overall and by pre-challenge status (ATP cohort for immunogenicity).
Table 3. Number and percentage of participants with anamnestic response to the challenge dose, overall and by pre-challenge status (ATP cohort for immunogenicity).
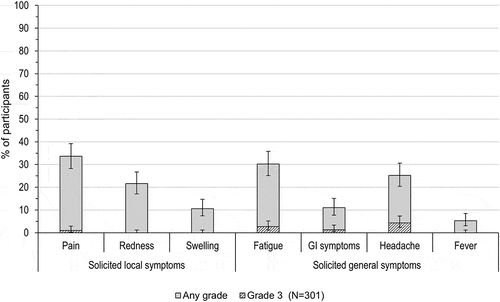
Figure 3. Percentage of adolescents with solicited local and general symptoms (total vaccinated cohort).
GI, gastrointestinal; Fever, temperature ≥ 37.5°C; N, number of participants with documented dose.Note: Grade 3 events were defined as preventing normal daily activities (for pain, fatigue, GI symptoms and headache), surface >50 mm (for redness, swelling) and temperature >39°C (fever). Error bars represent 95% confidence intervals.