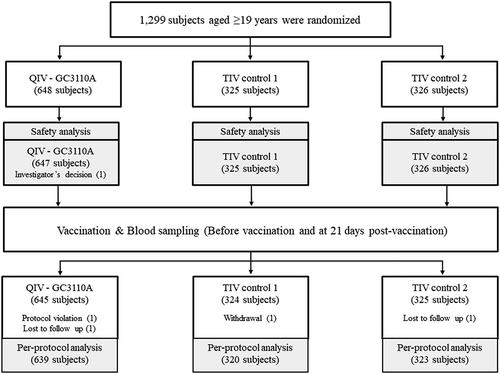
Figures & data
Table 1. Demographic characteristics of the study subjects.
Table 2. Post-vaccination non-inferiority analysis, as measured using a hemagglutination-inhibition (HI) assay (per-protocol population): trivalent influenza vaccines (controls) versus quadrivalent influenza vaccine (GC3110A).
Table 3. Post-vaccination immune responses, as measured using a hemagglutination-inhibition (HI) assay (per-protocol population): quadrivalent influenza vaccine (GC3110A) and trivalent influenza vaccines (controls).
Table 4. Solicited local and systemic adverse events within 21 days after vaccination: quadrivalent influenza vaccine (GC3110A) and trivalent influenza vaccines (controls).