Figures & data
Figure 1. Participant disposition. Primary series and booster data from Madhi et al (Study 1, South Africa)Citation16,Citation24 and Lopez et al (Study 2, Colombia) Citation20.
*, coadministered with MMR-V; †, coadministered with PCV7 (2, 4, 6 months) and rotavirus vaccine (2, 4 months); ‡, coadministered with PCV7 and MMR-V; #, data only available for Colombia at 3.5 years and 4.5 years of age in Study 2 (due to closure of site in Costa Rica)
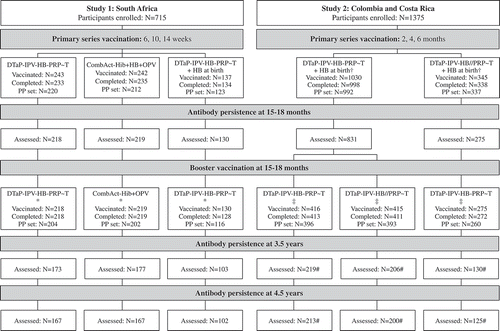
Table 1. Anti-hepatitis B antibody response post-primary vaccination, pre- and post-booster in second year of life, and persistence at 3.5 and 4.5 years of age.
Table 2. Anti-diphtheria antibody response post-primary vaccination, pre- and post-booster in second year of life, and persistence at 3.5 and 4.5 years of age.
Table 3. Anti-tetanus antibody response post-primary vaccination, pre- and post-booster in second year of life, and persistence at 3.5 and 4.5 years of age.
Table 4. Anti-poliovirus 1, 2, and 3 antibody response post-primary vaccination, pre- and post-booster in second year of life, and persistence at 3.5 and 4.5 years of age.
Table 5. Anti-pertussis (anti-PT and anti-FHA) antibody geometric mean concentrations post-primary vaccination, pre- and post-booster in second year of life, and persistence at 3.5 and 4.5 years of age.
Table 6. Anti-PRP antibody response post-primary vaccination, pre- and post-booster in second year of life, and persistence at 3.5 and 4.5 years of age.
