Figures & data
Figure 1. Application situation on mAb drugs clinical trials in China. Data source: www.yaozh.com.
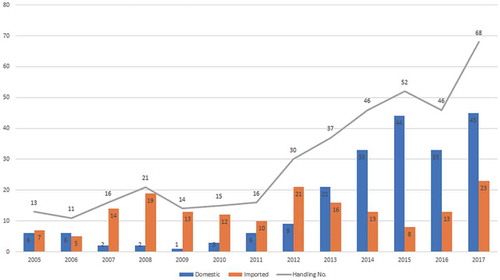
Figure 2. Registration situation on mAbs drugs clinical trials in China. Data source: Registration of drug clinical trials and information publicity platform.
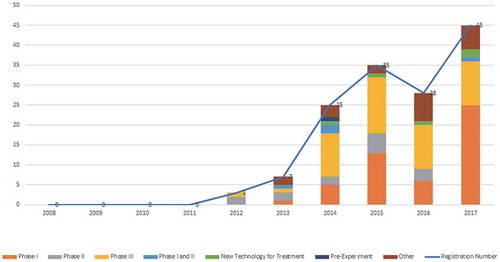
Figure 3. Research fields of mAb drugs clinical trials in China. Data source: Registration of drug clinical trials and information publicity platform.
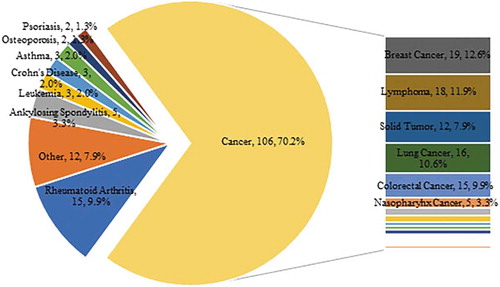
Table 1. Domestically approved mAb drugs by 2017.
Figure 4. Drug lag of mAb drugs approved by China compared with the countries that granted the first approval (by 2017).
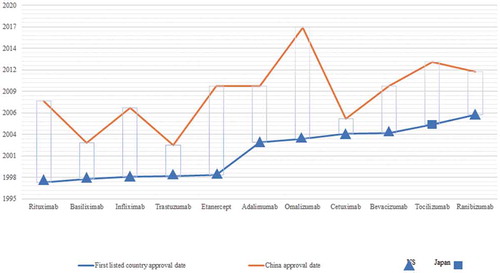
Table 2. Approval lag time of mAb drugs in different regions.
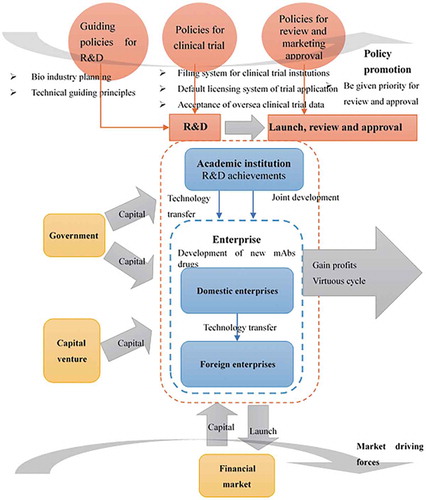
Table 3. Supports of R&D and guidance policies for Chinese biological drugs.
Table 4. China’s bio-pharmaceutical clinical trial policy.
Table 5. China bio-medicine marketing approval policy.