Figures & data
Figure 1. Flow diagram of search results and study selection.
ISPPD, International Symposium on Pneumococci and Pneumococcal Diseases; RCT, randomised controlled trial; SLR, systematic literature review.
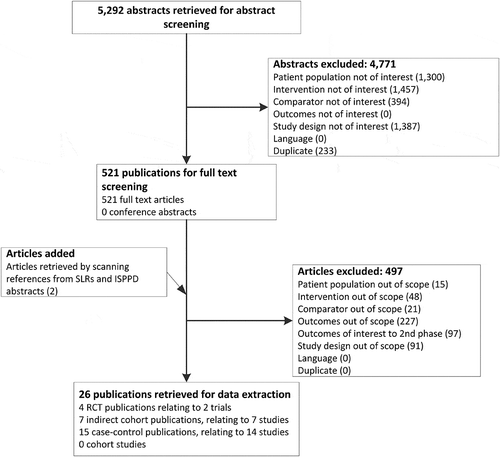
Table 1. Key study characteristics.
Figure 2. Summary of risk of bias assessment for (a) RCTs using the Cochrane risk of bias tool and (b) non-RCTs using the Newcastle-Ottawa scale.
RCT, randomized controlled trial.
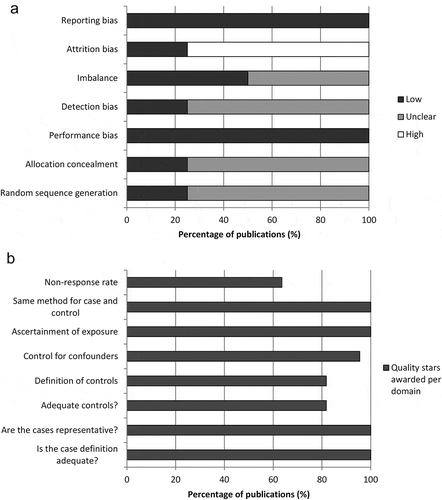
Figure 3. Overall network for included studies.
PCV-7, 7-valent pneumococcal conjugate vaccine; PCV-13, 13-valent pneumococcal conjugate vaccine; PHiD-CV, pneumococcal non-typeable Haemophilus influenzae protein D conjugate vaccine; RCT, randomized controlled trial. *It cannot be assumed that exposure is solely PCV-13
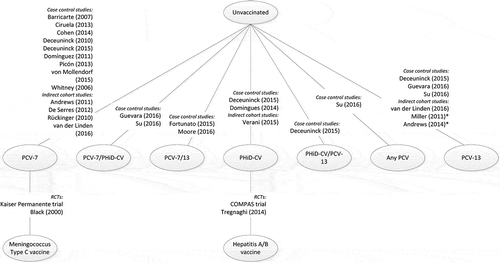
Table 2. Subject characteristics reported across studies.

