Figures & data
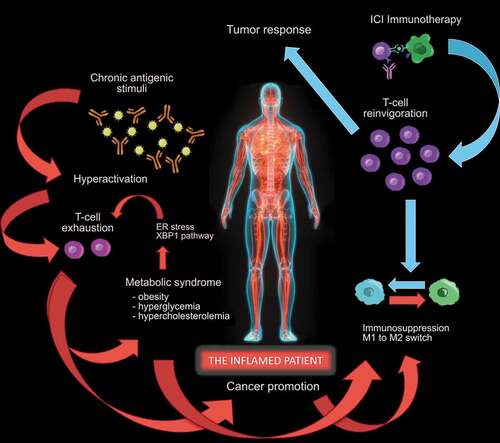
Figure 1. The “inflamed patient” is an individual whose immune system is dysfunctional, due to the exhaustion of T cells, that initially acquired effector functions, undergoing persistent antigen exposure. T-cell exhaustion is associated with inefficient control of inflammation and carcinogenesis, mediated by sustained upregulation of multiple inhibitory receptors (including PD-1), constituting the basis for promotion of cancer and metabolic syndrome, representing the epiphenomena of an inflamed patient. The manifestation of the metabolic syndrome includes obesity, hyperglycemia and hypercholesterolemia, the latter in turn favoring the CD8 + T-cell exhaustion in an endoplasmic-reticulum (ER)-stress-XBP1-dependent manner in the tumor microenvironment (TME). These phenomena trigger a potential vicious circle, rendering the patient even more “inflamed”, and favoring the M1 to M2 shift both in peripheral tissues and in the TME, furtherly diminishing T cell anti-tumor immune responses. The reversion of the initiating event, represented by T-cell exhaustion, can be reached with immune-checkpoint inhibitor (ICI) therapy, blocking the PD-1/PD-L1 axis and obtaining T-cell reinvigoration. This favors the recovery of the immune homeostasis, shifting the balance from immune-suppression to immune-functionality and promoting the T-cell response against the tumor