Figures & data
Table 1. Sequence of peptides used in the present study
Table 2. Immunization protocols of nanoparticle vaccine in C57BL/6 mice
Figure 1. Identification of biological features of nanoparticles
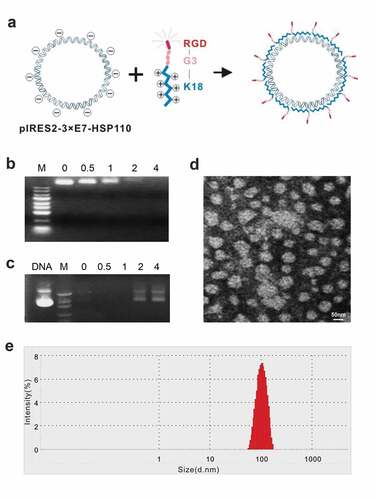
Figure 2. AK-nanoparticles targeted TC-1 cells and expressed exogenous protein
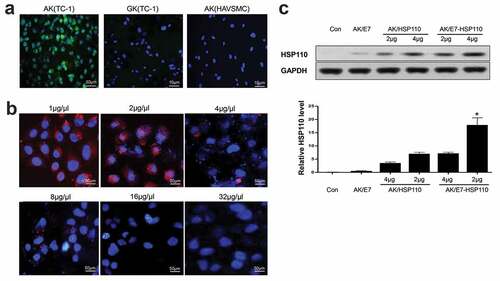
Figure 3. Nanoparticle AK/E7-HSP110 elicited E749-57-specific lymphocyte proliferation and cytolytic capacity
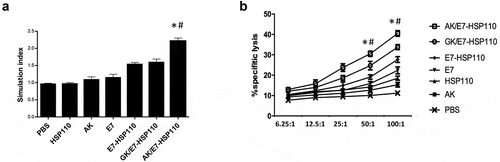
Figure 4. Specific CD8+T-cell immune response promoted by nanoparticle AK/E7-HSP110 in mice
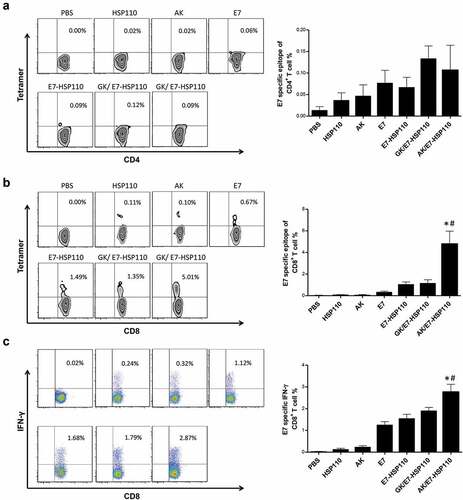
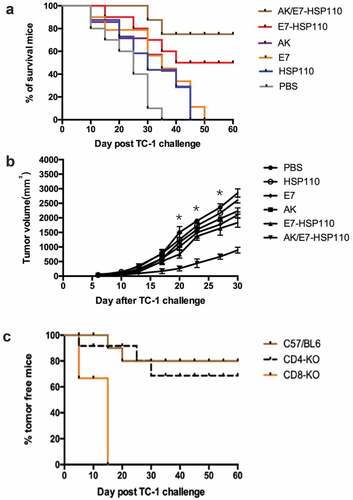
Figure 5. Preventative and therapeutic effects of vaccines on mice inoculated with TC-1 tumor cells