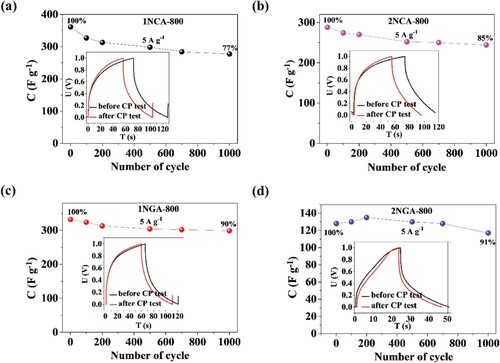
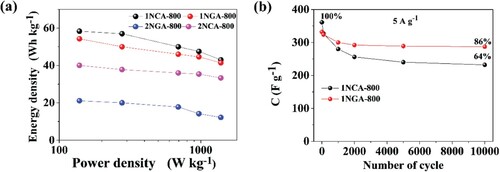
Figures & data

Figure 2. (a,b) Adsorption–desorption isotherms of N2 measured at −196°C on porous carbon materials. (c,d) Pore size distributions (PSDs) were obtained by the NLDFT method.
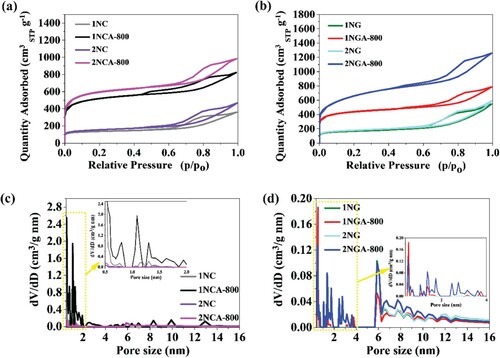
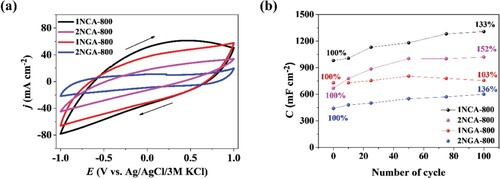
Figure 3. (a) Cyclic voltammetry curves recorded for carbon materials in 0.2 M K2SO4 (at a scan rate of v = 50 mV s−1) and (b) areal capacitance calculated on the basis of 100 galvanostatic charge/discharge cycles for carbon materials at a current density j = 3 mA cm−2.