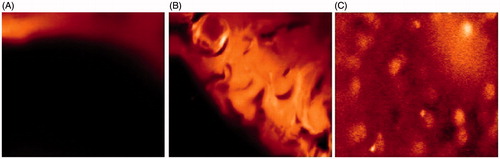
Figures & data
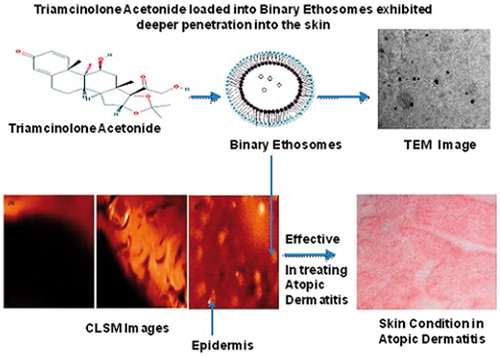
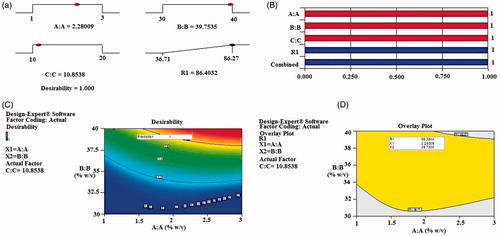
Table 1. Actual experiments with coded factor levels for optimization process and pharmacotechnical properties of EGVs.
Table 2. Formulation design and characterization for the preparation of triamcinolone acetonide (TA) loaded gels.
Figure 1. (A) In vitro release profiles of triamcinolone acetonide in phosphate buffer, pH 6.8 in 8 h using dialysis membrane; (B) In vitro cumulative permeability profiles of different gel formulations in phosphate buffer, pH 6.8.
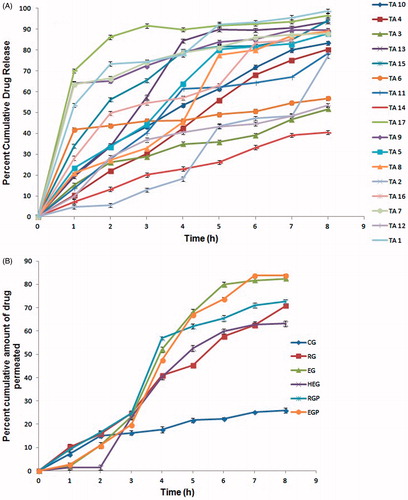
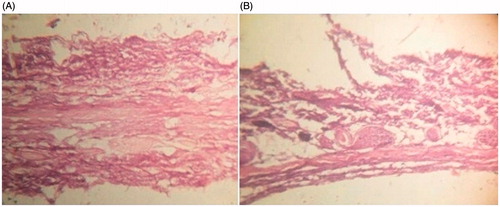
Figure 2. Transmission electron micrographs of (A) Reference ethosomes, RE; (B) Ethosomal glycolic vesicles, TA10.
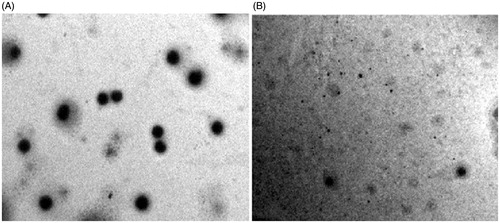
Table 3. Comparative stability investigation of optimized ethosomal glycolic vesicles (TA10) and reference ethosomes (RE).