Figures & data
Figure 1. Characterization of targeting molecular. (A) MALDI-TOF-MS spectrum of DSPE-PEG2000-NHS, (B) MALDI-TOF-MS spectrum of DSPE-PEG2000-OCT.
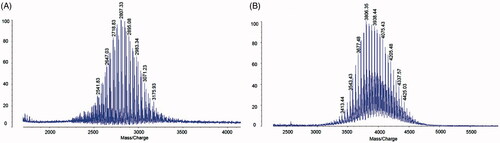
Figure 2. Characterization of OCT-modified daunorubicin plus dihydroartemisinin liposomes. (A) A schematic representation of OCT-modified daunorubicin plus dihydroartemisinin liposomes, (B) AFM image of OCT-modified daunorubicin plus dihydroartemisinin liposomes, (C) size distribution of OCT-modified daunorubicin plus dihydroartemisinin liposomes, (D) TEM of OCT-modified daunorubicin plus dihydroartemisinin liposomes.
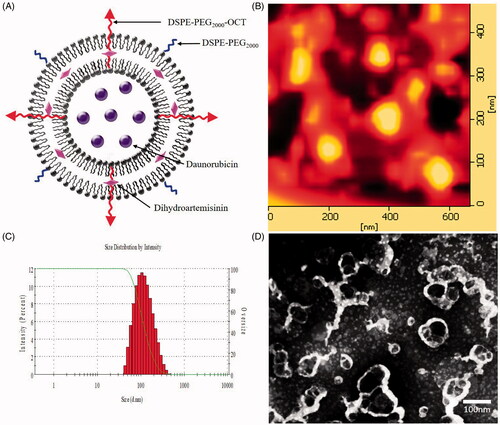
Table 1. Characterization of liposomes.
Figure 3. Inhibitory effects to breast cancer cells after treatment with OCT-modified daunorubicin plus dihydroartemisinin liposomes. p < .05; 1, versus free dihydroartemisinin; 2, versus free daunorubicin; 3, versus free daunorubicin plus 2.5 μM dihydroartemisinin liposomes; 4, versus free daunorubicin plus 5 μM dihydroartemisinin liposomes; a, versus blank liposomes; b, versus daunorubicin liposomes; c, versus daunorubicin plus dihydroartemisinin liposomes. Data are presented as mean ± SD (n = 6).
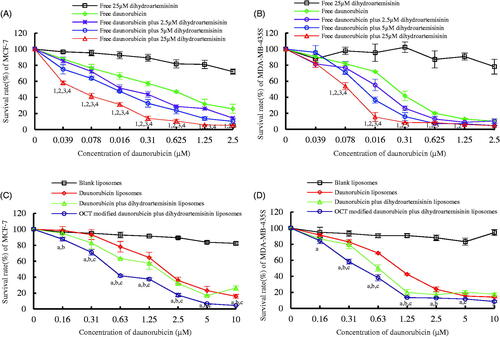
Table 2. IC50 values of daunorubicin (μM) against breast cancer cells.
Figure 4. Cellular uptake and targeting effects after incubation with varying formulations. (A) Cellular uptake of MCF-7 cells, (B) cellular uptake of MDA-MB-435S cells, (C) fluorescence microscopy images of MDA-MB-435S cells incubated with varying formulations. (a) Blank control; (b) daunorubicin liposomes; (c) OCT-modified daunorubicin plus dihydroartemisinin liposomes; (d) free daunorubicin.
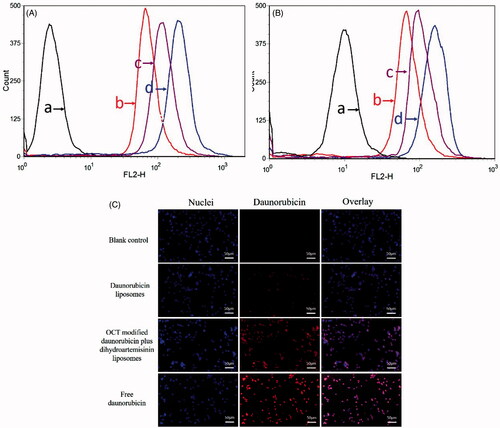
Figure 5. Blocking effects on MDA-MB-435S cells wound-healing and migration in vitro after treatment with varying formulations. (A) Blocking effects on MDA-MB-435S cells wound-healing; (B) blocking effects on MDA-MB-435S cells migration.
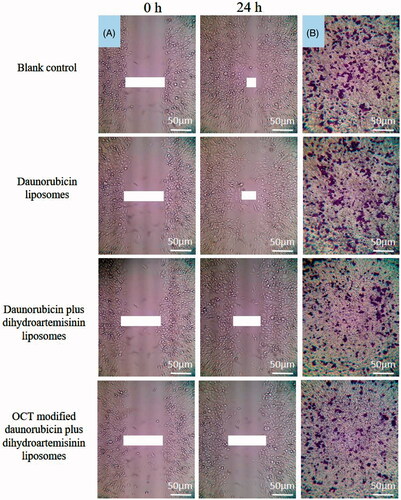
Figure 6. Regulating effects on the protein indicators of the EMT in MDA-MB-435S cells after treatments with OCT-modified daunorubicin plus dihydroartemisinin liposomes. (A) Blank control; (B) daunorubicin liposomes; (C) daunorubicin plus dihydroartemisinin liposomes; (D) OCT-modified daunorubicin plus dihydroartemisinin liposomes. p < .05; 1, versus A; 2, versus B; 3, versus C. Data are presented as mean ± SD (n = 4).
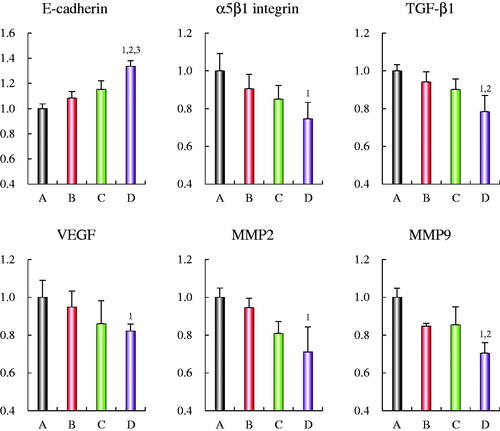
Figure 7. In vivo real-time imaging observation after intravenous administration of varying formulations.
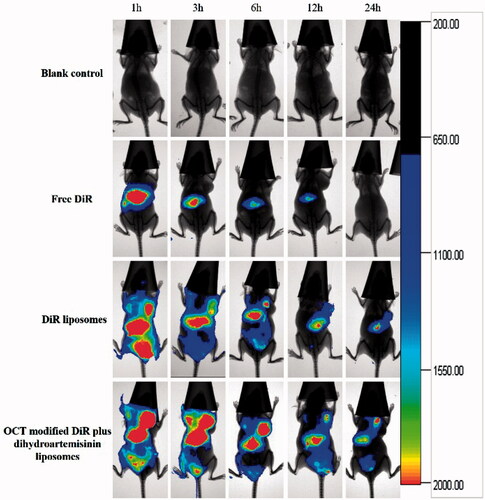
Figure 8. Anticancer efficacy on invasive breast cancer MDA-MB-435S cell xenografts nude mice after treatment with varying formulations. (A) Antitumor efficacy after treatment with varying formulations; (B) body weight changes during the treatment process. p < .05; 1, versus physiological saline. Data are presented as mean ± SD (n = 6).
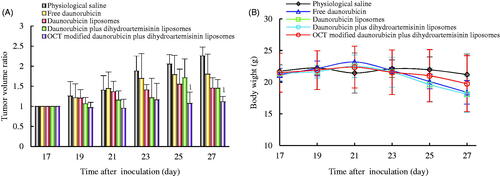
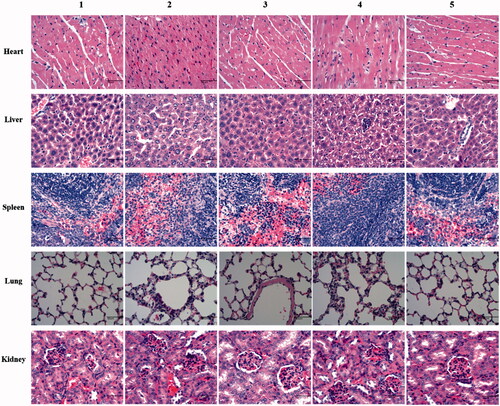
Figure 9. Histopathological analysis of main organs with H&E staining. (1) Physiological saline; (2) free daunorubicin; (3) daunorubicin liposomes; (4) daunorubicin plus dihydroartemisinin liposomes; (5) OCT-modified daunorubicin plus dihydroartemisinin liposomes. Images were obtained under 20× objectives.