Figures & data
Figure 1. XRD patterns presenting the phase of crystal structure of pure barium nanohexaferrites on varying the calcination conditions.
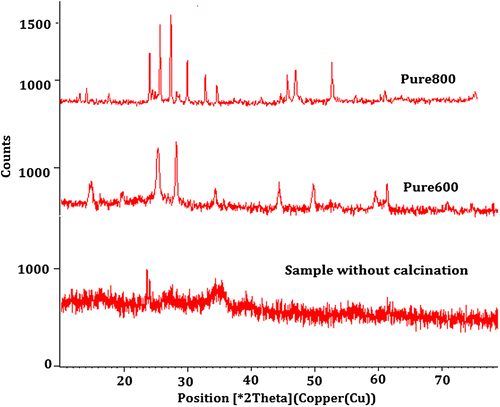
Figure 2. XRD patterns presenting the nickel-doped structure of pure barium nanohexaferrite with different values of nickel concentration.
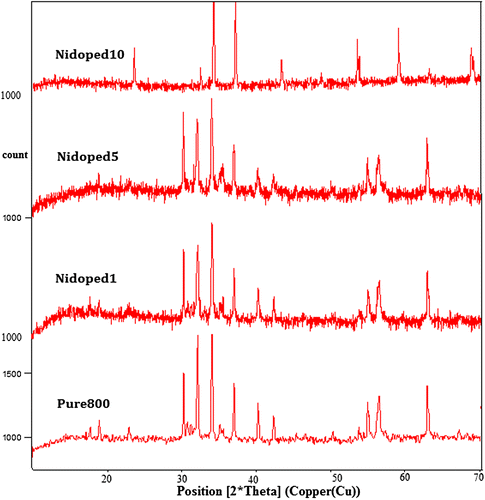
Table 1. Size calculation of synthesized samples for calcination duration of 4 h using Debye–Scherrer equation
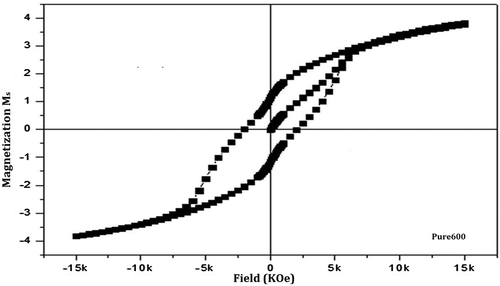
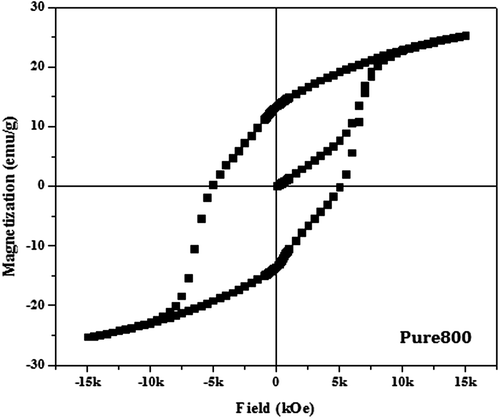
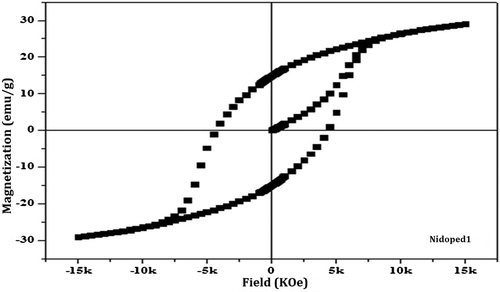
Table 2. Magnetic parameters of pure and doped barium nanohexaferrite samples
Figure 8. Real part of the relative permittivity of pure and nickel-doped barium nanohexaferrites as a function of logarithm of frequency.
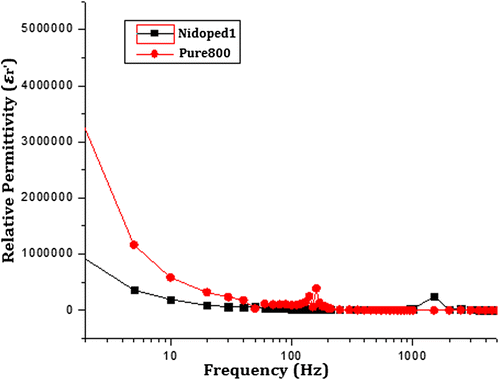
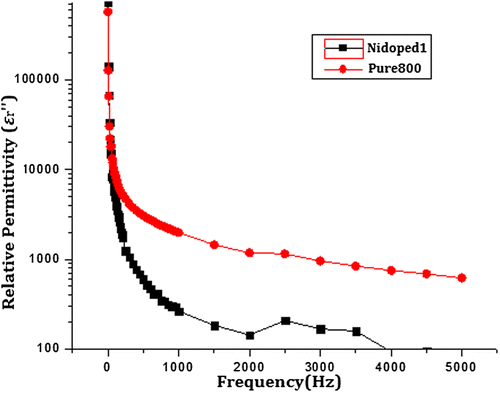
Figure 9. Imaginary part of relative permittivity of pure and nickel-doped barium nanohexaferrites as a function of frequency.