Figures & data
Table 1. Comparison of the proposed method with previously reported solvent extraction methods
Figure 1. (A) Plot of pH vs. percentage extraction of rhodium(III). Condition: Rhodium(III) = 200 μg, sodium malonate = 0.03 M, n-octylaniline = 0.1 M in toluene, shaking time = 3 min. (B) Extraction of rhodium(III) as a function of n-octylaniline concentration. Condition: Rhodium = 200 μg, pH = 9.0, sodium malonate = 0.03 M, shaking time = 3 min. (C) Extraction behavior of rhodium(III) as a function of weak organic acid concentration. Condition: Rhodium(III) = 200 μg, pH = 9.0, n-octylaniline = 0.1 M in toluene, shaking time = 3 min.
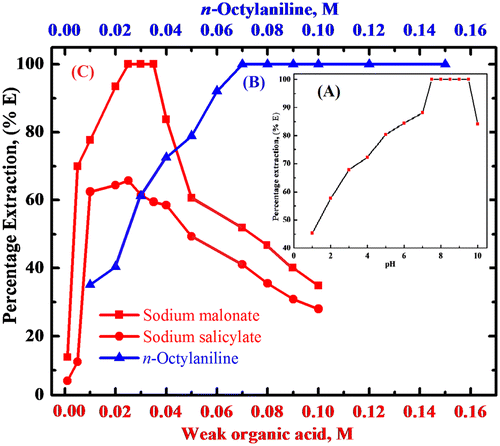
Table 2. Extraction behavior of rhodium(III) as a function of diluents
Table 3. Extraction behavior of rhodium(III) as a function of equilibrium time
Table 4. Extraction behavior of rhodium(III) as a function of stripping agents
Figure 2. (A) Log–log plot of Log D[Rh(III)] vs. Log C[malonate] at fixed n-octylaniline concentration. (B) Log–log plot of Log D[Rh(III)]] vs. Log C[n-octylaniline] at fixed malonate concentration.
![Figure 2. (A) Log–log plot of Log D[Rh(III)] vs. Log C[malonate] at fixed n-octylaniline concentration. (B) Log–log plot of Log D[Rh(III)]] vs. Log C[n-octylaniline] at fixed malonate concentration.](/cms/asset/83dfe674-9144-453b-aebc-dbec7a324064/oach_a_1339334_f0002_oc.gif)
